
��ҵ���û�ͭ��ұ��ͭ����¯���ۺ����õ�һ�ֹ����������£�
��1��ұ�������еõ�Cu2O��Cu�Ļ�����Ϊ����ͭ�����������A1�ڸ��������»�Ϸ�Ӧ�ɵô�ͭ����Ӧ��ѧ����ʽΪ________����ͭ����ʱӦ����ͭ������ֱ����Դ��____��������____���õ����Ƚϸߵľ�ͭ��
��2����ͳ��ͭ�ķ�����Ҫ�ǻ���ͭ������Ҫ��ӦΪ��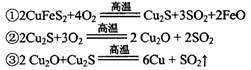
ÿ����1 mol Cu��������____mol O2����Ӧ���е���������____��
��3����ͭ������¯��(�� )���Ʊ�Fe2O3���������̻ش��������⣺
)���Ʊ�Fe2O3���������̻ش��������⣺
�ټ�������NaClO��Һ��Ŀ����_______ �������ӷ���ʽ��ʾ����
�ڳ�ȥAl3�������ӷ���ʽ��____��
��ѡ���ṩ���Լ������ʵ����֤¯���к���FeO���ṩ���Լ��У�ϡ���ᡢϡ���ᡢKSCN��Һ��KMnO4��Һ��NaOH��Һ����ˮ����ѡ�Լ���____��ʵ����ƣ�________��
��1��3Cu2O+2Al Al2O3+6Cu (2��) �� (1��) �� (1��)
Al2O3+6Cu (2��) �� (1��) �� (1��)
��2��2.5 (1��) Cu2O��Cu2S (1��)
��3����2Fe2++ClO-+2H+=2Fe3++Cl-+H2O (2��)
��Al3++4OH-=AlO2-+2H2O (2��)
��ϡ���� ���������Һ (2��)
ȡ�������������Һ���Թ��У��μӾ�ϡ�����ܽ��¯����Һ�����������Һ����ɫ��ȥ��֤������Fe2+ (2��)
���������������1���������֪������ͭ�������A1�ڸ��������»�Ϸ�Ӧ�ɵô�ͭΪ���ȷ�Ӧ����⾫��ͭ�Ǵ�ͭ�������������ӵ�Դ��������2������Ŀ����������ʽ������ɵã�2CuFeS2+5O2=2FeO+4SO2+2Cu��ÿ����1 mol Cu��������2.5mol O2������ͭ�Ļ��ϼ۽��ͣ���Cu2O��Cu2S����������3��¯���к���FeO����Ҫ�����������������л�ԭ�ԣ��ʼ���ʱ��ѡ�����Ը�����ء�
���㣺������ҵ���̿���������ԭ֪ʶ



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ʯ�ͺ�ú̿�ӹ������漰���ּ������豸��
(1)ʯ�ͷ���ʱ���ڲ�����________(�ҵ�豸����)��Ͷ��ԭ�ϵ�ͬʱ��ò�Ʒ���ù���Ϊ________�������̡�
(2)ʯ���ѻ���Ϊ���ѻ���________�ͼ����ѻ����ѻ���Ŀ�������________�IJ�����
(3)ú��ϴѡ��Ϊ�˽���ԭú�лҷֺ�________�ĺ�����ú��������ȼ����ָ�����ӵײ�����ú̿��������ʹȫ��ú̿����________����ȼ�յĹ��̡�
(4)ú��ֱ��Һ����ú���ʵ��ܼ���Ϻ��ڸ��º�________��������________��������Һ��ȼ�ϵĹ��̡�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ʯ�����ȼҵ�е�һ�ַ���������������±���ʾ��
�õ�ʯ����������ˮCaCl2��ij��������������¹������̣�
��֪�Ȼ��ƾ���Ļ�ѧʽ�ǣ�CaCl2��6H2O��H2S��һ���������壬�Ҿ��л�ԭ�ԡ�
��1����Ӧ���м������Ӧѡ��___________________��
��2����ɫ����Ӧ���������X��_______________���豸A��������______________���豸B������Ϊ________________���豸C��������____________________��
��3��Ϊ�����㻷��Ҫ���轫����H2Sͨ�����ճأ��������������ʺ���Ϊ���ռ�����_____________����Ӧ�Ļ�ѧ����ʽΪ_________________��
| A��ˮ | B��Ũ���� | C��ʯ���� | D������ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
������أ�K2FeO4����һ�ּ�������������������һ������Ͷ��ˮ���������������������£�
��֪����2KOH + Cl2 =" KCl" + KClO + H2O���������¶Ƚϵͣ�
��6KOH + 3Cl2 =" 5KCl" + KClO3 + 3H2O���������¶Ƚϸߣ�
��1������������Ӧ�� ����¶Ƚϸߡ����¶Ƚϵ͡�������½��У�
��2��д����ҵ����ȡCl2�Ļ�ѧ����ʽ ��
��3���ڡ���ӦҺI���м�KOH�����Ŀ���� ��
��4��������������Һ���ɷ�ӦҺ��Ļ�ѧ����ʽ��
��5���ӡ���ӦҺ���з����K2FeO4���и���Ʒ ��д��ѧʽ�������Ƕ�����Ҫ�Ļ�����Ʒ������˵������һ�����ʵ���; ��
��6��������K2FeO4�ܽ���pH��4.74����Һ�У����Ƴ�c(FeO42��) ��1.0 mmol��L��1���������������ֱ�����20�桢30�桢40���60��ĺ���ˮԡ�У��ⶨc(FeO42��)�ı仯�������ͼ����ʵ���Ŀ���� ��������Ӧ�ġ�H 0�����������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�������ѣ�TiO2���㷺Ӧ���ڸ���ṹ����Ϳ�ϡ�ֽ��Ϳ��ȣ��������ѻ�����Ϊ�Ʊ��ѵ��ʵ�ԭ�ϡ�
�������ѿ����������ַ����Ʊ���
����1��TiCl4ˮ������TiO2��xH2O�����ˡ�ˮϴ��ȥ���е�Cl�����ٺ�ɡ����ճ�ȥˮ�ֵõ�����TiO2���˷����Ʊ��õ��������������ѡ�
��1���� TiCl4ˮ������TiO2��x H2O�Ļ�ѧ����ʽΪ_______________________________ ��
�ڼ���TiO2��x H2O��Cl���Ƿ����ķ�����______________________________
����2�����ú���Fe2O3����������Ҫ�ɷ�ΪFeTiO3������TiԪ�ػ��ϼ�Ϊ+4�ۣ���ȡ������Ҫ�������£�
��2���������ĩ�м����ᷴӦ��TiO2+�����ӷ���ʽΪ
��3����Һ���м���Fe��������
��4����Ҫ����FeSO4��7H2O�������ˮ�������������ƾ��ơ��������⣬��Ҫ�õ������ֹ�������������
��������ѿ�������ȡ�ѵ��ʣ��漰���IJ�������ͼ��
��Ӧ�ڵķ���ʽ�� ���÷�Ӧ��Ҫ��Ar�����н��У������ԭ�� _
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
���������ƣ�Na2S2O5��������ʳƷƯ�������Ʊ������������£�
��֪����Ӧ�����2NaHSO3 Na2S2O5��H2O�ȶಽ��Ӧ��
Na2S2O5��H2O�ȶಽ��Ӧ��
��1��ʵ������ȡ�����Ļ�ѧ����ʽ�� ��
��2����ӦI�Ļ�ѧ����ʽΪ�� ��
��3�������ա�ʱ������Ӧ�Ļ�ѧ����ʽ�� ��
��4����֪Na2S2O5��ϡ���ᷴӦ�ų�SO2�������ӷ���ʽΪ�� ��
��5������ƷX�Ļ�ѧʽ�� ��
��6��Ϊ�˼��ٲ�ƷNa2S2O5�����ʺ���������Ʒ�Ӧ�����������������ʵ���֮��ԼΪ �������Ʒ�к���̼�������������Լ��� �����ţ�
�����Ը������ ��Ʒ����Һ �۳���ʯ��ˮ
�ܱ���̼��������Һ ��NaOH ��ϡ����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��п��������������ηɻ��������״ﲨͿ�ϡ����ԷϾ�п�̵��Ϊԭ���Ʊ���п���������Ҫ�������£�
(1)���ʱ���������̱�˫��ˮ��ԭ�Ļ�ѧ����ʽΪ_______��
(2)�������۳���ʱ�����۵�������______ (�����������ԭ������������������
(3)�������Ե���Ϊ����������Һ������������KMnO4��Һ�������ն�ʵ�ֵġ�����ͼ��KMnO4��Һ���ڲ�ͬpHʱ��Ӧ��Hgȥ���ʱ仯ͼ��ͼ������ΪHg�� MnO4-�ڸ�pH��Χ�ڵ���Ҫ���
������ݸ�ͼ����pH�Թ�ȥ����Ӱ��ı仯���ɣ�______��
���Բ�����ǿ���Ի����¹��ĵ�λʱ��ȥ���ʸߵ�ԭ��______��
(4)��x��0.2ʱ�����õ�����п��������״ﲨ�����������ر�ǿ���������������ʽ��ʾ�����������____________��
(5)���ⶨ��Һ�ɷֺ�ͬʱ����һ������MnSO4�����۵�Ŀ����______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����Ǧ(PbSO4)�㷺Ӧ�����������ء���ɫ���ϵȡ�����пұ�������е�Ǧ��������PbSO4���������£�
��֪Ǧ��������Ҫ�ɷ���PbO��Pb������������Ag��Zn��CaO��������������������ʡ�25��ʱ��Ksp(CaSO4)=4.9x10-5��Ksp(PbSO4)=1.6xl0��8��
��1����֪����I��NO�������������Һ�к���������������Pb2+���ֱ�д��PbO��Pb�μӷ�Ӧ�Ļ�ѧ����ʽ ��
��2������I����������������ʹPb����ʣ�ܣ�Ŀ���� ��
��3��ĸҺ��ѭ�������ڲ���I����������Ҫ�� (��һ�����ʻ�ѧʽ������ĸҺ�в�����SO42�����࣬ѭ������ʱ���ܳ��ֵ������� ��
��4����ƷPbSO4������Pb(NO3)2��Һ���ϴ�ӣ�Ŀ���dz�ȥ ��
��5��Ǧ���صĵ��Һ�����ᣬ���������缫�ϳ�����PbSO4�ֱ�ת��ΪPbO2��Pb�����ʱ�����ĵ缫��ӦʽΪ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
����ʵ�ʲ����ڹ�ҵ�������� (����)��
| A��CO2ͨ������������Һ����Na2CO3 |
| B��H2��Cl2������HCl |
| C��Cl2ͨ�����ʯ��ˮ����Ư�� |
| D������������� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com