
ĀČ»ÆĢśŹĒ³£¼ūµÄĖ®“¦Ąķ¼Į£¬ĄūÓĆ·ĻĢśŠ¼æÉÖʱøĪŽĖ®ĀČ»ÆĢś”£ŹµŃéŹŅÖʱø×°ÖĆŗĶ¹¤ŅµÖʱøĮ÷³ĢĶ¼ČēĻĀ£ŗ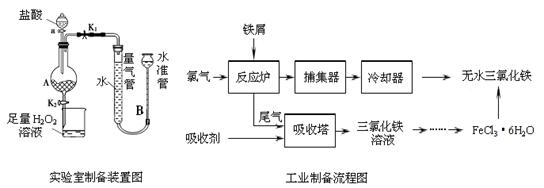
ŅŃÖŖ£ŗ(1)ĪŽĖ®FeCl3µÄČŪµćĪŖ555 K”¢·ŠµćĪŖ588 K”£
(2) ·ĻĢśŠ¼ÖŠµÄŌÓÖŹ²»ÓėŃĪĖį·“Ó¦
(3)²»Ķ¬ĪĀ¶ČĻĀĮłĖ®ŗĻĀČ»ÆĢśŌŚĖ®ÖŠµÄČܽā¶ČČēĻĀ£ŗ
| ĪĀ¶Č/”ę | 0 | 20 | 80 | 100 |
| Čܽā¶Č(g/100 g H2O) | 74.4 | 91.8 | 525.8 | 535.7 |
£Ø1£©°ŃŃĒĢśĄė×ÓČ«²æŃõ»Æ³ÉČż¼ŪĢśĄė×Ó £Ø2·Ö£©
£Ø2£©×°ÖĆAÖŠ²»²śÉśĘųÅŻ»ņĮæĘų¹ÜŗĶĖ®×¼¹ÜŅŗĆę²»±ä£ØĘäĖūŗĻĄķ“š°øŅ²øų·Ö£©£Ø2·Ö£©
£Ø3£©ŃĪĖį£Ø1·Ö£© Õō·¢ÅØĖõ”¢ĄäČ“½į¾§£Ø1·Ö£©
£Ø4£©2Fe2+ + Cl2 = 2Fe3+ £Ø2·Ö£©
£Ø5£©Fe2Cl6£Ø2·Ö£©
£Ø6£©¢Ł ČÜŅŗÓÉĄ¶É«±äĪŽÉ«£¬ĒŅ°ė·ÖÖÓÄŚ²»±äÉ«£Ø2·Ö£©
¢Ś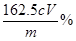 £Ø2·Ö£©
£Ø2·Ö£©
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©øł¾ŻŹµŃé×°ÖĆŗĶ²Ł×÷²½ÖčæÉÖŖ£¬ŹµŃéŹŅÖʱøĪŽĖ®FeCl3£¬ŹĒĄūÓĆĢśÓėŃĪĖį·“Ӧɜ³ÉFeCl2£¬Č»ŗó°ŃFeCl2Ńõ»Æ³ÉFeCl3£¬ĻČÖʵĆFeCl3”¤6H2O¾§Ģ壬ŌŁĶŃ½į¾§Ė®ÖʵĆĪŽĖ®FeCl3”£ÉÕ±ÖŠ×ćĮæµÄH2O2ČÜŅŗŹĒ×÷Ńõ»Æ¼Į£¬°ŃŃĒĢśĄė×ÓČ«²æŃõ»Æ³ÉČż¼ŪĢśĄė×Ó£»
£Ø2£©ĢśÓėŃĪĖį·“Ó¦ĶźČ«Ź±£¬²»ŌŁ²śÉśĒāĘų£¬ĖłŅŌ×°ÖĆAÖŠ²»²śÉśĘųÅŻ»ņĮæĘų¹ÜŗĶĖ®×¼¹ÜµÄŅŗĆę²»ŌŁ±ä»Æ£¬“ĖŹ±£¬æɽ«AÖŠFeCl2ČÜŅŗ·ÅČėÉÕ±ÖŠ½ųŠŠŃõ»Æ”£
£Ø3£©“ÓFeCl3ČÜŅŗÖʵĆFeCl3?6H2O¾§ĢåµÄ²Ł×÷²½ÖčŹĒ£ŗÕō·¢ÅØĖõ”¢ĄäČ“½į¾§”¢¹żĀĖ”¢Ļ“µÓ”¢øÉŌļ£®
£Ø4£©“Ó¹¤ŅµÖʱøĮ÷³ĢĶ¼·ÖĪöæÉÖŖ£¬·“Ó¦ĀÆÖŠ½ųŠŠµÄ·“Ó¦ŹĒ 2Fe + 3Cl2 = 2FeCl3£¬Ņņ“Ė£¬½ųČėĪüŹÕĖžÖŠµÄĪ²ĘųŹĒƻӊ²Ī¼Ó·“Ó¦µÄĀČĘų£¬ŌŚĪüŹÕĖžÖŠĀČĘų±»ĪüŹÕ¼ĮĪüŹÕ£¬·“Ó¦ŗóÉś³É2FeCl3ČÜŅŗ£¬ĖłŅŌĪüŹÕ¼ĮÓ¦ŹĒ2FeCl2ČÜŅŗ£¬·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ£ŗ2Fe2+ + Cl2 = 2Fe3+”£
£Ø5£©²¶¼ÆĘ÷ŹÕ¼ÆµÄŹĒĘųĢ¬FeCl3£¬FeCl3µÄĻą¶Ō·Ö×ÓÖŹĮæŹĒ162.5£¬ÓÉĻą¶Ō·Ö×ÓÖŹĮæĪŖ325µÄĢśµÄĀČ»ÆĪļæÉŅŌĶĘ³ö£¬µ±ĪĀ¶Č³¬¹ż673 KŹ±£¬¶ž·Ö×ÓĘųĢ¬FeCl3æÉŅŌ¾ŪŗĻÉś³ÉĖ«¾ŪĢåFe2Cl6”££Ø±ø×¢£ŗFeCl3Źō¹²¼ŪŠĶ»ÆŗĻĪļ£¬ÄÜČÜÓŚŅŅĆŃ”¢±ūĶŖµČÓŠ»śČܼĮ£¬ŌŚ673KÕōĘųÖŠÓŠĖ«¾ŪĢ壬1023KŅŌÉĻ·Ö½āĪŖµ„·Ö×Ó£©”£
£Ø6£©¢Ł³ĘČ”m gĪŽĖ®ĀČ»ÆĢśŃłĘ·£¬ČÜÓŚĻ”ŃĪĖį£¬ÅäÖĘ³É100mLČÜŅŗ£»Č”³ö10.00mL£¬¼ÓČėÉŌ¹żĮæµÄKIČÜŅŗ£¬³ä·Ö·“Ó¦ŗ󣬵ĪČė¼øµĪµķ·ŪČÜŅŗ£¬“ĖŹ±ČÜŅŗ³ŹĄ¶É«£¬ÓĆ Na2S2O3ČÜŅŗµĪ¶Ø£¬µĪČė×īŗóŅ»µĪNa2S2O3ČÜŅŗ£¬×¶ŠĪĘæÄŚµÄČÜŅŗÓÉĄ¶É«±äĪŖĪŽÉ«£¬ĒŅ°ė·ÖÖÓÄŚ²»±äÉ«”£ĖłŅŌµĪ¶ØÖÕµćµÄĻÖĻóŹĒ£ŗČÜŅŗÓÉĄ¶É«±äĪŖĪŽÉ«£¬ĒŅ°ė·ÖÖÓÄŚ²»±äÉ«”£
¢Ś Ēóѳʷ֊ĀČ»ÆĢśµÄÖŹĮæ·ÖŹż
ÓÉ·“Ó¦£ŗ2Fe3+ + 2I - = 2Fe2+ + I2£» I2+2S2O32-ØT2I-+S4O62-£»æÉµĆ¹ŲĻµŹ½£ŗ
2Fe3+ ”Ŗ I2 ”Ŗ 2S2O32-
1 1
n cV”Į10-3
ĒóµĆn(Fe3+) = cV”Į10-3 mol£¬ Ōņѳʷ֊ĀČ»ÆĢśµÄÖŹĮæ·ÖŹżĪŖ£ŗ
¦Ų(Fe3+) = cV”Į10-3 mol”Į10”Į162.5g/mol”Į100% = 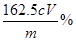
æ¼µć£ŗ»ÆѧŹµŃéÉč¼ĘÓėĘĄ¼Ū


 Ķ¬²½Į·Ļ°ŗÓÄĻ“óѧ³ö°ęÉēĻµĮŠ“š°ø
Ķ¬²½Į·Ļ°ŗÓÄĻ“óѧ³ö°ęÉēĻµĮŠ“š°ø Ķ¬²½Į·Ļ°Ī÷ÄĻŹ¦·¶“óѧ³ö°ęÉēĻµĮŠ“š°ø
Ķ¬²½Į·Ļ°Ī÷ÄĻŹ¦·¶“óѧ³ö°ęÉēĻµĮŠ“š°ø ²¹³äĻ°Ģā½ĖÕĻµĮŠ“š°ø
²¹³äĻ°Ģā½ĖÕĻµĮŠ“š°ø ѧĮ·æģ³µµĄæŚĖćŠÄĖćĖŁĖćĢģĢģĮ·ĻµĮŠ“š°ø
ѧĮ·æģ³µµĄæŚĖćŠÄĖćĖŁĖćĢģĢģĮ·ĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĪŹ“šĢā
£Ø14·Ö£©Īų34SeŗĶķŚ52Te¶¼ŹĒµŚVIA×åŌŖĖŲ£¬ĪųŹĒ·Ö²¼ŌŚµŲæĒÖŠµÄĻ”ÓŠŌŖĖŲ”£¹¤ŅµÉĻÓĆĪų¹Ä·ĻĮĻ£ØÖ÷ŅŖ³É·ÖĪų”¢ķŚ”¢Ģ¼”¢ĶŗĶĢśŗĻ½š£©»ŲŹÕ¾«Į¶ĪųµÄĮ÷³ĢČēĻĀ£ŗ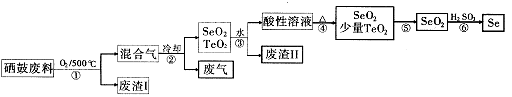
ŅŃÖŖ£ŗ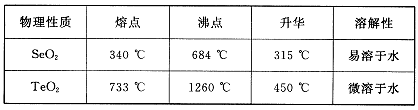
£Ø1£©SeµÄĒā»ÆĪļµÄµē×ÓŹ½ŹĒ____”£
£Ø2£©²½Öč¢ŁÖŠĶØČĖµÄŃõĘųŹ¹Īų¹Ä·ĻĮĻ·ĢŚ£¬ÄæµÄŹĒ ”£
£Ø3£©·ĻĘųµÄÖ÷ŅŖ³É·ÖŹĒ____ £»·ĻŌüIIµÄÖ÷ŅŖ³É·ÖŹĒ ”£
£Ø4£©²½Öč¢ÜÖŠÖ÷ŅŖ·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ £»²½Öč¢Ž·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ ”£
£Ø5£©øł¾Ż±ķÖŠŹż¾Ż£¬²½Öč¢Ż×īŹŹŅĖµÄ·ÖĄė·½·ØŹĒ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
ÓĆŗ¬ÓŠA12O3”¢SiO2ŗĶÉŁĮæFeO”¤xFe2O3µÄĀĮ»ŅÖʱøA12(SO4)3”¤18H2O£¬¹¤ŅÕĮ÷³ĢČēĻĀ£Ø²æ·Ö²Ł×÷ŗĶĢõ¼žĀŌ£©
¢ń£®ĻņĀĮ»ŅÖŠ¼ÓČė¹żĮæĻ”H2SO4£¬¹żĀĖ:
¢ņ£®ĻņĀĖŅŗÖŠ¼ÓČė¹żĮæKMnO4ČÜŅŗ£¬µ÷½ŚČÜŅŗµÄpHŌ¼ĪŖ3£»
¢ó£®¼ÓČČ£¬²śÉś“óĮæ×ŲÉ«³Įµķ£¬¾²ÖĆ£¬ÉĻ²ćČÜŅŗ³Ź×ĻŗģÉ«:
¢ō£®¼ÓČėMnSO4ÖĮ×ĻŗģÉ«ĻūŹ§£¬¹żĀĖ;
¢õ£®ÅØĖõ”¢½į¾§”¢·ÖĄė£¬µĆµ½²śĘ·”£
£Ø1£©H2SO4ČܽāA12O3µÄĄė×Ó·½³ĢŹ½ŹĒ
£Ø2£©KMnO4Ńõ»ÆFe2+µÄĄė×Ó·½³ĢŹ½²¹³äĶźÕū£ŗ
£Ø3£©ŅŃÖŖ£ŗÉś³ÉĒāŃõ»ÆĪļ³ĮµķµÄpH
| | Al(OH)3 | Fe(OH)2 | Fe(OH)3 |
| æŖŹ¼³ĮµķŹ± | 3.4 | 6.3 | 1.5 |
| ĶźČ«³ĮµķŹ± | 4.7 | 8.3 | 2.8 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
°¢Ė¾Ę„ĮÖ£ØŅŅõ£Ė®ŃīĖį£¬ £©ŹĒŹĄ½ēÉĻÓ¦ÓĆ×ī¹ć·ŗµÄ½āČČ”¢ÕņĶ“ŗĶæ¹Ń×Ņ©”£ŅŅõ£Ė®ŃīĖįŹÜČČŅ×·Ö½ā£¬·Ö½āĪĀ¶ČĪŖ128”«135”ę”£Ä³Ń§Ļ°Š”×éŌŚŹµŃéŹŅŅŌĖ®ŃīĖį£ØĮŚōĒ»ł±½¼×Ėį£©Óė“×Ėįōū[(CH3CO)2O]ĪŖÖ÷ŅŖŌĮĻŗĻ³É°¢Ė¾Ę„ĮÖ£¬Öʱø»ł±¾²Ł×÷Į÷³ĢČēĻĀ£ŗ
£©ŹĒŹĄ½ēÉĻÓ¦ÓĆ×ī¹ć·ŗµÄ½āČČ”¢ÕņĶ“ŗĶæ¹Ń×Ņ©”£ŅŅõ£Ė®ŃīĖįŹÜČČŅ×·Ö½ā£¬·Ö½āĪĀ¶ČĪŖ128”«135”ę”£Ä³Ń§Ļ°Š”×éŌŚŹµŃéŹŅŅŌĖ®ŃīĖį£ØĮŚōĒ»ł±½¼×Ėį£©Óė“×Ėįōū[(CH3CO)2O]ĪŖÖ÷ŅŖŌĮĻŗĻ³É°¢Ė¾Ę„ĮÖ£¬Öʱø»ł±¾²Ł×÷Į÷³ĢČēĻĀ£ŗ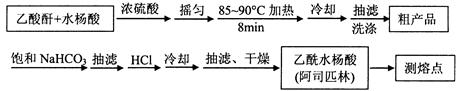
Ö÷ŅŖŹŌ¼ĮŗĶ²śĘ·µÄĪļĄķ³£Źż
Ēėøł¾ŻŅŌÉĻŠÅĻ¢»Ų“šĻĀĮŠĪŹĢā£ŗ
£Ø1£©¢ŁŗĻ³É°¢Ė¹Ę„ĮÖŹ±£¬×īŗĻŹŹµÄ¼ÓČČ·½Ź½ŹĒ ”£
¢Ś³éĀĖĖłµĆ“Ö²śĘ·ŅŖÓĆÉŁĮæ±łĖ®Ļ“µÓ£¬ŌņĻ“µÓµÄ¾ßĢå²Ł×÷ŹĒ ”£
£Ø2£©Ģį“æ“Ö²śĪļÖŠ¼ÓČė±„ŗĶNaHCO3ČÜŅŗÖĮƻӊCO2²śÉśĪŖÖ¹£¬ŌŁ³éĀĖ£¬Ōņ¼Ó±„ŗĶNaHCO3ČÜŅŗµÄÄæµÄŹĒ ”£
ĮķŅ»ÖÖøĽųµÄĢį“æ·½·Ø£¬³ĘĪŖÖŲ½į¾§Ģį“æ·Ø”£·½·ØČēĻĀ£ŗ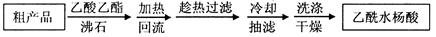
£Ø3£©øĽųµÄĢį“æ·½·ØÖŠ¼ÓČČ»ŲĮ÷µÄ×°ÖĆČēĶ¼ĖłŹ¾£¬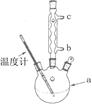
¢ŁŹ¹ÓĆĪĀ¶Č¼ĘµÄÄæµÄŹĒ ”£ĄäÄżĖ®µÄĮ÷½ų·½ĻņŹĒ ”££ØĢī”°b”±»ņ”°c”±£©”£
¢ŚÖŲ½į¾§Ģį“æ·ØĖłµĆ²śĘ·µÄÓŠ»śŌÓÖŹŅŖ±ČŌ·½°øÉŁµÄŌŅņ ”£
£Ø4£©¼ģŃé²śĘ·ÖŠŹĒ·ńŗ¬ÓŠĖ®ŃīĖįµÄ»Æѧ·½·ØŹĒ ”£
£Ø5£©øĆѧĻ°Š”×éŌŚŹµŃéÖŠŌĮĻÓĆĮæ£ŗ2.0 gĖ®ŃīĖį”¢5.0 mL“×Ėįōū£Ø¦Ń£½1.08 g/cm3)£¬×īÖÕ³ĘµĆ²śĘ·m£½2.2 g£¬ŌņĖłµĆŅŅõ£Ė®ŃīĖįµÄ²śĀŹĪŖ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
ĄūÓĆĖį½ā·ØÖĘīŃ°×·Ū²śÉśµÄ·ĻŅŗ[ŗ¬ÓŠ“óĮæFeSO4”¢H2SO4ŗĶÉŁĮæFe2(SO4)3TiOSO4]£¬Éś²śĮņĖįŃĒĢśŗĶ²¹ŃŖ¼ĮČéĖįŃĒĢś”£ĘäÉś²ś²½ÖčČēĻĀ£ŗ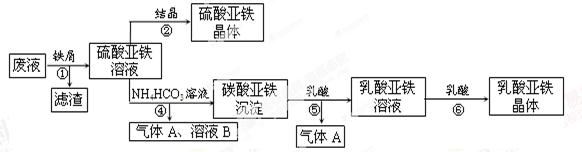
Ēė»Ų“š£ŗ
£Ø1£©²½Öč¢ŁÖŠ·ÖĄė²Ł×÷µÄĆū³ĘŹĒ______£»²½Öč¢Ž±ŲŠėæŲÖĘŅ»¶ØµÄÕęæÕ¶Č£¬ŌŅņŹĒ_________£»
£Ø2£©·ĻŅŗÖŠµÄTiOSO4ŌŚ²½Öč¢ŁÄÜĖ®½āÉś³ÉĀĖŌü£ØÖ÷ŅŖ³É·ÖĪŖTiO2?xH2O£©µÄ»Æѧ·½³ĢŹ½ĪŖ__________£»²½Öč¢ÜµÄĄė×Ó·½³ĢŹ½ĪŖ_____________________”£
£Ø3£©ÓĆĘ½ŗāŅʶÆŌĄķ½āŹĶ²½Öč¢ŻÖŠ¼ÓČéĖįÄܵƵ½ČéĖįŃĒĢśµÄŌŅņ_______”£
£Ø4£©ÅäĘ½ĖįŠŌøßĆĢĖį¼ŲČÜŅŗÓėĮņĖįŃĒĢśČÜŅŗ·“Ó¦µÄĄė×Ó·½³ĢŹ½£ŗ_____Fe2++_____MnO4£+_____H+=_____Fe3++_____Mn2++_____
Č”²½Öč¢ŚĖłµĆ¾§Ģåѳʷag,ČÜÓŚĻ”ĮņĖįÅä³É100.00 mLČÜŅŗ£¬Č”³ö20. 00 mLČÜŅŗ,ÓĆKMnO4ČÜŅŗµĪ¶Ø£ØŌÓÖŹÓėKMnO4²»·“Ó¦£©”£ČōĻūŗÄ0.1000 mol”¤L£1KMnO4ČÜŅŗ20.00mL£¬ŌņĖłµĆ¾§ĢåÖŠFeSO4?7H2OµÄÖŹĮæ·ÖŹżĪŖ£ØŅŌŗ¬aµÄŹ½×Ó±ķŹ¾£©_____________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
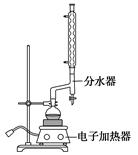
äå±½ŹĒŅ»Öֻƹ¤ŌĮĻ£¬ŹµŃéŹŅŗĻ³Éäå±½µÄ×°ÖĆŹ¾ŅāĶ¼¼°ÓŠ¹ŲŹż¾ŻČēĻĀ£ŗ
| | ±½ | äå | äå±½ |
| ĆܶČ/(g”¤cm£3) | 0.88 | 3.10 | 1.50 |
| ·Šµć/”ę | 80 | 59 | 156 |
| Ė®ÖŠČܽā¶Č | Ī¢ČÜ | Ī¢ČÜ | Ī¢ČÜ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
µāŹĒČĖĢå±ŲŠčµÄŌŖĖŲÖ®Ņ»,ŗ£ŃóÖ²ĪļČēŗ£“ų”¢ŗ£ŌåÖŠŗ¬ÓŠ·įø»µÄ”¢ŅŌ»ÆŗĻĢ¬ŠĪŹ½“ęŌŚµÄµāŌŖĖŲ”£ŌŚŹµŃéŹŅÖŠ,“Óŗ£ŌåĄļĢįČ”µāµÄĮ÷³ĢŗĶŹµŃé×°ÖĆČēĻĀ: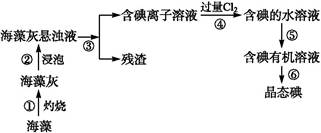
(1)Öø³öÉĻŹöĢįČ”µāµÄ¹ż³ĢÖŠÓŠ¹ŲŹµŃé²Ł×÷µÄĆū³Ę:²½Öč¢Ū””””””””,²½Öč¢Ż”””””””””£
(2)Š“³ö²½Öč¢Ü¶ŌÓ¦·“Ó¦µÄĄė×Ó·½³ĢŹ½:”””””””””””””””””””””£
(3)ĢįČ”µāµÄ¹ż³ĢÖŠ,æɹ©Ń”ŌńµÄÓŠ»śŹŌ¼ĮŹĒ”””””””””£(Ģī±ąŗÅ)
A.¾Ę¾« B.“×Ėį C.ĖÄĀČ»ÆĢ¼ D.±½
(4)²½Öč¢Ü³żĮĖ¼ÓČė¹żĮæCl2,ĻĀĮŠŃõ»Æ¼Į×īŗĆŃ”ÓĆ””””””””(Ģī“śŗÅ)”£
A.ÅØĮņĖį B.H2O2ČÜŅŗ C.KMnO4ČÜŅŗ
ĄķÓÉŹĒ____________________________”£
(5)ĪŖĮĖŹ¹ŗ£Ōå»ŅÖŠµÄµāĄė×Ó×Ŗ»ÆĪŖµāµÄÓŠ»śČÜŅŗ,¼“Ķź³É²½Öč¢ŪÖĮ¢Ż,ŹµŃéŹŅĄļÓŠÉÕ±”¢²£Į§°ō”¢¼ÆĘųĘ攢¾Ę¾«µĘ”¢µ¼¹Ü”¢Ō²µ×ÉÕĘ攢ŹÆĆŽĶųŅŌ¼°±ŲŅŖµÄ¼Š³ÖŅĒĘ÷ŗĶĪļĘ·,ɊȱɣµÄ²£Į§ŅĒĘ÷ŹĒ”””””””””£
(6)“Óŗ¬µāµÄÓŠ»śČܼĮÖŠĢįČ”µāŗĶ»ŲŹÕÓŠ»śČܼĮ,»¹ŠčŅŖ¾¹żÕōĮó”£Öø³öĻĀĶ¼ŹµŃé×°ÖĆÖŠ“ęŌŚµÄ“ķĪóÖ®“¦:”””£ 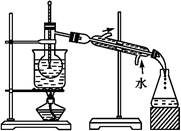
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
ijĶ¬Ń§Éč¼ĘĮĖČēĶ¼ĖłŹ¾×°ÖĆ½ųŠŠŹÆÓĶ·ÖĮóµÄŹµŃ锣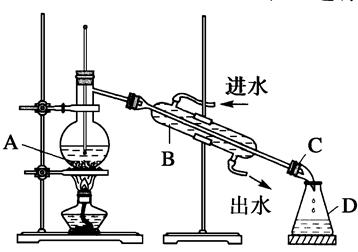
£Ø1£©Öø³öŹµŃé×°ÖĆÖŠŅĒĘ÷A”¢B”¢C”¢DµÄĆū³Ę£ŗ
A________ӢB________ӢC________ӢD________ӣ
£Ø2£©Öø³öøĆĶ¬Ń§ĖłÉč¼ĘµÄŹµŃé×°ÖĆÖŠ“ęŌŚµÄ“ķĪ󣬲¢øųÓčøÄÕż”£
“ķĪó£ŗ¢Ł_______________________________________£»
¢Ś_____________________________________________”£
øÄÕż£ŗ¢Ł_______________________________________£»
¢Ś_____________________________________________”£
£Ø3£©ŹµŃé×°ÖĆøÄÕżŗ󣬽ųŠŠĘųĆÜŠŌ¼ģ²éµÄ·½·Ø£ŗ______________________________”£
£Ø4£©ÕōĮóÉÕĘæÖŠ·ÅČė¼øʬĖé“ÉʬµÄ×÷ÓĆŹĒ__________________________________”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŹµŃéĢā
±½¼×ĖįŅŅõ„£ØC9H10O2£©ÉŌÓŠĖ®¹ūĘųĪ¶£¬ÓĆÓŚÅäÖĘĻćĖ®Ļć¾«ŗĶČĖŌģ¾«ÓĶ£¬“óĮæÓĆÓŚŹ³Ę·¹¤ŅµÖŠ£¬Ņ²æÉÓĆ×÷ÓŠ»śŗĻ³ÉÖŠ¼äĢ唢ČܼĮµČ”£ĘäÖʱø·½·ØĪŖ£ŗ
ŅŃÖŖ£ŗ
| | ŃÕÉ«”¢×“Ģ¬ | ·Šµć£Ø”ę£© | ”” ĆÜ¶Č£Øg”¤cm£3£© |
| *±½¼×Ėį | ĪŽÉ«”¢Ę¬×“¾§Ģå | 249 | 1.2659 |
| ±½¼×ĖįŅŅõ„ | ĪŽÉ«³ĪĒåŅŗĢå | 212.6 | 1.05 |
| ŅŅ“¼ | ĪŽÉ«³ĪĒåŅŗĢå | 78.3 | 0.7893 |
| »·¼ŗĶé | ĪŽÉ«³ĪĒåŅŗĢå | 80.8 | 0.7318 |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com