
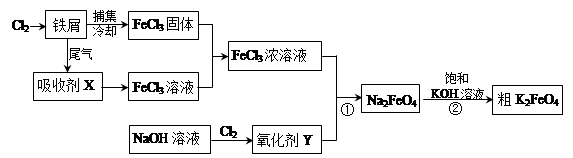
 4Fe(OH)3+8OH-+3O2”ü”£ŌŚĢį“æK2FeO4Ź±²ÉÓĆÖŲ½į¾§”¢Ļ“µÓ”¢µĶĪĀŗęøɵķ½·Ø£¬ŌņĻ“µÓ¼Į×īŗĆŃ”ÓĆ £ØĢīŠņŗÅ£©”£
4Fe(OH)3+8OH-+3O2”ü”£ŌŚĢį“æK2FeO4Ź±²ÉÓĆÖŲ½į¾§”¢Ļ“µÓ”¢µĶĪĀŗęøɵķ½·Ø£¬ŌņĻ“µÓ¼Į×īŗĆŃ”ÓĆ £ØĢīŠņŗÅ£©”£| A£®H2O | B£®Ļ”KOHČÜŅŗ”¢Ņģ±ū“¼ | C£®NH4ClČÜŅŗ”¢Ņģ±ū“¼ | D£®Fe(NO3)3ČÜŅŗ”¢Ņģ±ū“¼ |
 CrO42-£«Fe(OH)3”ż£«OH-
CrO42-£«Fe(OH)3”ż£«OH- Cr2O72-£«H2O
Cr2O72-£«H2O 2Cr3£«£«6Fe3£«£«7H2O
2Cr3£«£«6Fe3£«£«7H2O Fe(OH)3(½ŗĢå) + 3H+£Ø2·Ö£©
Fe(OH)3(½ŗĢå) + 3H+£Ø2·Ö£© Fe(OH)3(½ŗĢå) + 3H+”£
Fe(OH)3(½ŗĢå) + 3H+”£ 4Fe(OH)3+8OH-+3O2”üæÉÖŖ£¬Ģį“æK2FeO4Ó¦øĆŅÖÖĘøĆ·“Ó¦£¬ĖłŅŌ×īŗƵďŌ¼ĮŹĒĻ”KOHČÜŅŗ£ØŅÖÖĘĘ½ŗā£©”¢Ņģ±ū“¼£Ø¼õŠ”Čܽā£©£¬“š°øŃ”B”£
4Fe(OH)3+8OH-+3O2”üæÉÖŖ£¬Ģį“æK2FeO4Ó¦øĆŅÖÖĘøĆ·“Ó¦£¬ĖłŅŌ×īŗƵďŌ¼ĮŹĒĻ”KOHČÜŅŗ£ØŅÖÖĘĘ½ŗā£©”¢Ņģ±ū“¼£Ø¼õŠ”Čܽā£©£¬“š°øŃ”B”£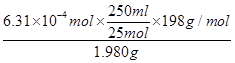 ”Į100%£½63£®1%
”Į100%£½63£®1%


| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
| ŠņŗÅ | µēŃ¹/V | Ńō¼«ĻÖĻó | ¼ģŃéŃō¼«²śĪļ |
| I | x”Ża | µē¼«ø½½ü³öĻÖ»ĘÉ«£¬ÓŠĘųÅŻ²śÉś | ÓŠFe3+”¢ÓŠCl2 |
| II | a£¾x”Żb | µē¼«ø½½ü³öĻÖ»ĘÉ«£¬ĪŽĘųÅŻ²śÉś | ÓŠFe3+”¢ĪŽCl2 |
| III | b£¾x£¾0 | ĪŽĆ÷ĻŌ±ä»Æ | ĪŽFe3+”¢ĪŽCl2 |
| ŠņŗÅ | µēŃ¹/V | Ńō¼«ĻÖĻó | ¼ģŃéŃō¼«²śĪļ |
| IV | a£¾x”Żc | ĪŽĆ÷ĻŌ±ä»Æ | ÓŠCl2 |
| V | c£¾x”Żb | ĪŽĆ÷ĻŌ±ä»Æ | ĪŽCl2 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®¢Ū¢Ż | B£®¢Ś¢Ż | C£®¢Ł¢Ś¢Ü | D£®¢Ł¢Ś¢Ū¢Ü¢Ż |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®ĻȵĪ¼ÓĀČĖ®£¬ŌŁµĪ¼ÓKSCNČÜŅŗŗóĻŌŗģÉ« |
| B£®ĻȵĪ¼ÓKSCNČÜŅŗ£¬²»ĻŌŗģÉ«£¬ŌŁµĪ¼ÓĀČĖ®ŗóĻŌŗģÉ« |
| C£®µĪ¼ÓNaOHČÜŅŗ£¬ĻČ²śÉś°×É«³Įµķ£¬ŗó±äĪŖ»ŅĀĢÉ«£¬×īŗó³ŹŗģŗÖÉ« |
| D£®Ö»ŠčµĪ¼ÓKSCNČÜŅŗ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
| A£®ŠæĶŌµē³ŲÖŠĶŹĒÕż¼« | B£®ÓƵē½ā·Ø¾«Į¶ĶŹ±“ÖĶ×÷Ņõ¼« |
| C£®ŌŚ¶Ę¼žÉĻ¶ĘĶŹ±ĶÓėµēŌ“Õż¼«ĻąĮ¬ | D£®Ķ×÷Ńō¼«Ź±²»Ņ»¶ØČܽā |
|
 ”¾ŹµŃé¹ż³Ģ”æ
”¾ŹµŃé¹ż³Ģ”æ²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®·½·Ø¢ŪŹĒ×ī¾¼ĆŗĶ»·±£µÄ |
| B£®·½·Ø¢ŁÖŠ£¬Čō“Ó¾¼ĆŗĶ»·±£µÄ½Ē¶Čæ¼ĀĒ£¬ÓĆĻ”ĻõĖį±ČÓĆÅØĻõĖįŗĆ |
| C£®·½·Ø¢ŚŠčŅŖĻūŗÄÄÜŌ“£¬¶Ō»·¾³²»²śÉśĪŪČ¾ |
| D£®·½·Ø¢ŪÖŠN2O4¼ČŹĒŃõ»Æ¼ĮÓÖŹĒ»¹Ō¼Į |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

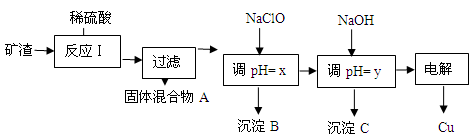
| ³ĮµķĪļ | Cu(OH)2 | Al(OH)3 | Fe(OH)3 | Fe(OH)2 |
| æŖŹ¼³ĮµķpH | 5.4 | 4.0 | 1.1 | 5.8 |
| ³ĮµķĶźČ«pH | 6.7 | 5.2 | 3.2 | 8.8 |
 HClO£¬ClO£ĻūŗÄH+£¬“Ó¶ų“ļµ½µ÷½ŚpHµÄÄæµÄ
HClO£¬ClO£ĻūŗÄH+£¬“Ó¶ų“ļµ½µ÷½ŚpHµÄÄæµÄ HClO+OH££¬OH£ĻūŗÄH+ £¬“Ó¶ų“ļµ½µ÷½ŚpHµÄÄæµÄ
HClO+OH££¬OH£ĻūŗÄH+ £¬“Ó¶ų“ļµ½µ÷½ŚpHµÄÄæµÄ²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
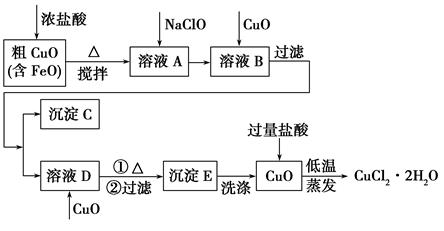
| ĪļÖŹ | Fe(OH)2 | Cu(OH)2 | Fe(OH)3 |
| ĶźČ«³ĮµķŹ±µÄpH | ”Ż9.6 | ”Ż6.4 | 3”«4 |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com