

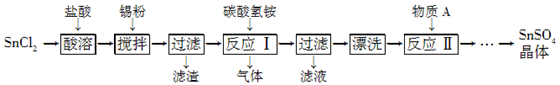
 Ca2£«+
Ca2£«+ ClO£+
ClO£+ SO2+
SO2+ H2O£½
H2O£½ ”ż+
”ż+ Cl£+
Cl£+ SO42£+
SO42£+ ”£
ӣ


| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĪŹ“šĢā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
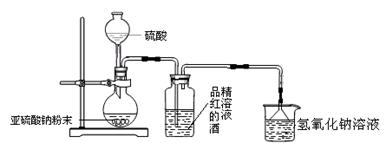
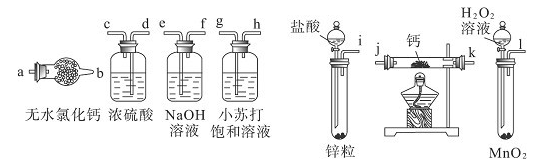
| A£®ŹµŃéŹŅÖʱøŅŅĻ©Ź±£¬²śÉśµÄĘųĢåŹ¹äåĖ®ĶŹÉ«£¬ĖµĆ÷ÓŠŅŅĻ©Éś³É |
| B£®½«Ņŗä唢Ģś·ŪŗĶ±½»ģŗĻ¼ÓČČ¼“æÉÖʵĆäå±½£¬³żČ„äå±½ÖŠŗģ×ŲÉ«µÄä壬æÉÓĆĻ”NaOHČÜŅŗ·“ø“Ļ“µÓ£¬²¢ÓĆ·ÖŅŗĀ©¶··ÖŅŗ |
| C£®ŌŚŅ»Ö§ŹŌ¹ÜÖŠµĪČė10µĪäåŅŅĶ飬ŌŁ¼ÓČė1mL5%µÄNaOHČÜŅŗ£¬¹²ČČŗóµĪ¼ÓĻõĖįŅųČÜŅŗ£¬æɹŪ²ģµ½ÓŠĒ³»ĘÉ«äå»ÆŅų³ĮµķÉś³É |
| D£®ÖĘČ”Ļõ»ł±½Ź±£¬Ó¦Č”ÅØH2SO42mL£¬¼ÓČė1.5mLÅØHNO3£¬ŌŁµĪČė±½Ō¼1mL£¬Č»ŗó·ÅŌŚĖ®Ō”ÖŠ¼ÓČČ£¬ĪĀ¶Č¼ĘÓ¦·ÅŌŚ»ģŗĻŅŗÖŠ |
²éæ““š°øŗĶ½āĪö>>
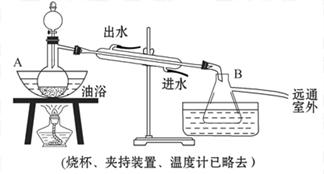
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
²éæ““š°øŗĶ½āĪö>>
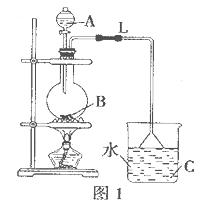
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
| A£®¼ÓČČ·“Ó¦Ņ»¶ĪŹ±¼ä | B£®ŹÕ¼ÆĘųĢå²¢¼ģŃéĘä“æ¶Č |
| C£®¹Ų±Õ·ÖŅŗĀ©¶·»īČū | D£®Ķ£Ö¹¼ÓČČ£¬³ä·ÖĄäČ“ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĪŹ“šĢā
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com