
ʵ������Na2CO3��������250 mL 0.1 mol/L��Na2CO3��Һ������д���пհף�
��1��ʵ��ʱ��Ӧ��������ƽ��ȡNa2CO3���� g��
��2��������Na2CO3��Һʱ���õIJ���������Ҫ����Ͳ���ձ����������� ��
��
��3��ʵ���������õ��������������÷ֱ���___________��__________��
��4����ʵ��������������������������������ҺŨ��ƫ�͵���_____��������ţ�
| A��ת��ǰ������ƿ�к�����������ˮ |
| B��ת��ʱ����������Һ���� |
| C������ʱ�����ӿ̶��� |
| D������ʱ�����ӿ̶��� |
��1��2.7��2�֣�
��2��250 mL����ƿ����ͷ�ιܣ�2�֣�
��3�����衢������2�֣�
��4��BC��3�֣�
���������������1��m=M��n=M��C��V=106g/mol��0.1 mol/L��0.25L=2.7g
��2����3��������Na2CO3��Һʱ��1����.2����.3�ܽ�.4ת��.5ϴ��.6����.7ҡ��8������Һ�����õIJ���������Ҫ����Ͳ���ձ���������������ƿ����ͷ�ιܣ����в������������ǽ��裬������
��4����A��Ӱ�졣D����ʱ�����ӿ̶���ʹ������ʵ��ƫ�ͣ�ʵ��Ũ�ȴ��ڶ�������ƫ�͡�
���㣺һ�����ʵ���Ũ�ȵ���Һ������
������һ�����ʵ���Ũ�ȵ�����ʱҪע�⣺1����.2����.3�ܽ�.4ת��.5ϴ��.6����.7ҡ��8������Һ.�Լ�Ҫ��Ǹ����Ĺؼ��㼰����Ҫ�㡣


 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010������ʡ������У�������һ��ѧ�����п��Ի�ѧ�� ���ͣ������
��13�֣�ʵ������Na2CO3��10H2O��������240mL 0.1mol/L Na2CO3��Һ���ش��������⣺
��1����������ƽ��ȡNa2CO3��10H2O������Ϊ g��
��2������������������������ƽ���ձ����������⣬����Ҫ �����������ƣ���
��3������ʱ����ȷ�IJ���˳���ǣ�����ĸ��ʾ��ÿ����ĸֻ����һ�Σ� ��
| A��������ˮϴ���ձ���������2-3�Σ�ϴ��Һ��ע������ƿ���� |
| B����������ƽ���������Na2CO3��10H2O���壬�����ձ��У��ټ�������ˮ���ò���������������ʹ����ȫ�ܽ� |
| C��������ȴ��Na2CO3��Һ�ز�����ע������ƿ�� |
| D��������ƿ�ǽ�����ҡ�� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013����ʡ�����и�һ��ѧ�����п��Ի�ѧ�Ծ��������棩 ���ͣ�ʵ����
ʵ������Na2CO3��������250 mL 0.1 mol/L��Na2CO3��Һ������д���пհף�
��1��ʵ��ʱ��Ӧ��������ƽ��ȡNa2CO3���� g��
��2��������Na2CO3��Һʱ���õIJ���������Ҫ����Ͳ���ձ����������� ��
��
��3��ʵ���������õ��������������÷ֱ���___________��__________��
��4����ʵ��������������������������������ҺŨ��ƫ�͵���_____��������ţ�
A��ת��ǰ������ƿ�к�����������ˮ
B��ת��ʱ����������Һ����
C������ʱ�����ӿ̶���
D������ʱ�����ӿ̶���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2015�������и�һ10���¿���ѧ�Ծ��������棩 ���ͣ�ʵ����
��14�֣�I��ʵ������Na2CO3��10H2O��������50 g ��������Ϊ21.2����Na2CO3��Һ���ش��������⣺
��1��Ӧ��������ƽ��ȡNa2CO3��10H2O���� g��
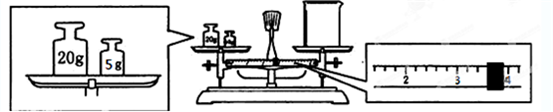
��2����������ƽ��С�ձ��Ƴ�̼���ƾ������������ƽƽ����״̬����ͼ����ͼ�п��Կ�������ͬѧ�ڲ���ʱ����һ�������� ��ʵ�ʳ�����̼���ƾ�������Ϊ g��

II. ʵ����Ҫ����2.5 mol/L ��ϡ������Һ90 mL���ش��������⣺
��1������Ͳ��ȡ��������Ϊ98�����ܶ�Ϊ1.84 g/cm3��Ũ���� mL��
��2������ʱ������ʹ�õ���������Ͳ���ձ����������⣬��ȱ�ٵ������� ��
��3��������Һ�Ĺ����У�������������ȷ�����в�����ʹ������ҺŨ��ƫ�ߵ��� ��
A����ȡŨ����ʱ�����Ӷ���
B��ϴ����ȡŨH2SO4�����Ͳ������ϴ��Һת�Ƶ�����ƿ��
C��ϡ������ʱ������Һ����������
D��û��ϴ��ϡ��������ձ��Ͳ�����
E������ҡ�Ⱥ���Һ����ڱ��ߣ����ý�ͷ�ιܼ�����ˮ������
F������ƿ������
��4��������ƿ��ȡ����Һ40 mL����5 mol/L��NaOH��Һ mLǡ����ȫ��Ӧ����Ӧ����Һ�е�c(Na��)�� ��������Һ��Ϲ����е�����仯��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com