

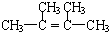
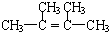




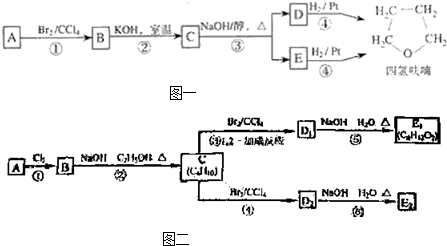
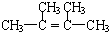 ����BΪ
����BΪ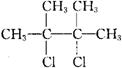 �����Ҵ��ļ�����Һ�м���ʱ������ȥ��Ӧ�����ɵ�CΪ
�����Ҵ��ļ�����Һ�м���ʱ������ȥ��Ӧ�����ɵ�CΪ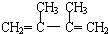 ��C����1��2-�ӳ�����D1����D1Ϊ
��C����1��2-�ӳ�����D1����D1Ϊ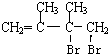 ��ˮ������E1��E1Ϊ
��ˮ������E1��E1Ϊ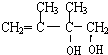 ��D1��D2��Ϊͬ���칹�壬��C����1��4-�ӳ�����D2����D2Ϊ
��D1��D2��Ϊͬ���칹�壬��C����1��4-�ӳ�����D2����D2Ϊ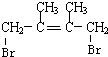 ��ˮ������E2��E2Ϊ
��ˮ������E2��E2Ϊ ���ݴ˽��
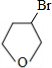
���ݴ˽��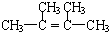 ����BΪ
����BΪ �����Ҵ��ļ�����Һ�м���ʱ������ȥ��Ӧ�����ɵ�CΪ
�����Ҵ��ļ�����Һ�м���ʱ������ȥ��Ӧ�����ɵ�CΪ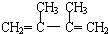 ��C����1��2-�ӳ�����D1����D1Ϊ
��C����1��2-�ӳ�����D1����D1Ϊ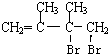 ��ˮ������E1��E1Ϊ
��ˮ������E1��E1Ϊ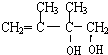 ��D1��D2��Ϊͬ���칹�壬��C����1��4-�ӳ�����D2����D2Ϊ
��D1��D2��Ϊͬ���칹�壬��C����1��4-�ӳ�����D2����D2Ϊ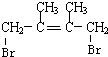 ��ˮ������E2��E2Ϊ
��ˮ������E2��E2Ϊ ��
��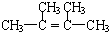 ���ʴ�Ϊ��
���ʴ�Ϊ��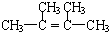 ��
�� ��CΪ
��CΪ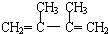 ����ѧ����Ϊ��2��3-����-1��3-����ϩ��������������֪��E2�Ľṹ��ʽ��
����ѧ����Ϊ��2��3-����-1��3-����ϩ��������������֪��E2�Ľṹ��ʽ�� ���ܵķ�Ӧ�����Ǽӳɷ�Ӧ��
���ܵķ�Ӧ�����Ǽӳɷ�Ӧ�� ��2��3-����-1��3-����ϩ��
��2��3-����-1��3-����ϩ�� ���ӳɷ�Ӧ��
���ӳɷ�Ӧ��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
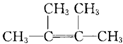
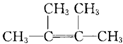

 +2NaOH
+2NaOH| CH3CH2OH |
| �� |
 +2NaCl+2H2O
+2NaCl+2H2O +2NaOH
+2NaOH| CH3CH2OH |
| �� |
 +2NaCl+2H2O
+2NaCl+2H2O�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| KOH/���� |





 +NaOH
+NaOH| ˮ |
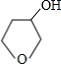 +NaBr
+NaBr +NaOH
+NaOH| ˮ |
 +NaBr
+NaBr| �� |
| �� |
| �� |
| �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�





�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com