
����Ŀ����ʢ�е����Ͷ����������������Թ�(�Թ��ݻ�ΪVL)������ˮ�У������㹻����ʱ����Թ�������������ΪaL����ش��������⣺
(1)�û�����嵹����ˮ�з�����Ӧ�Ļ�ѧ����ʽ��______________________��
(2)��a��![]() V����ԭ��������е�����������������֮����__________��
V����ԭ��������е�����������������֮����__________��
(3)��ϵ�(2)С�⣬��aL������ͨ���������Թ��иպ�ֻ��һ������ʱ��ֹͣͨ��������
���Թ�������ʣ���������____________���������____________��
�ڸù������ܷ�Ӧ�Ļ�ѧ����ʽ��__________________��
���𰸡�3NO2��H2O=2HNO3��NO1��3N![]() NO��3O2��2H2O=4HNO3
NO��3O2��2H2O=4HNO3
��������
(1)��������ж���������ˮ��Ӧ���������һ������������ʽΪ��3NO2��H2O=2HNO3��NO����2�����ݷ�Ӧǰ����������������㡣������嵹����ˮ�к��������![]() VL���ɿ�֪�������������Ϊ
VL���ɿ�֪�������������Ϊ![]() ��
��![]() VL��
VL��![]() VL�����������ΪVL��
VL�����������ΪVL��![]() VL��
VL��![]() VL��ԭ������嵪����������������Ϊ1��3����3��aLԭ�����������ˮʱͨ������ʱ��4NO��3O2��2H2O===4HNO3�����ʣ��һ������Ϊ�����������Ϊ
VL��ԭ������嵪����������������Ϊ1��3����3��aLԭ�����������ˮʱͨ������ʱ��4NO��3O2��2H2O===4HNO3�����ʣ��һ������Ϊ�����������Ϊ![]() V��
V��


 ������ʱͬ����ϰ��ϵ�д�
������ʱͬ����ϰ��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ж�һ����ѧ��Ӧ���Է��Գ������оݺ����оݣ�������������£������ж���Ӧһ�����Է����е��ǣ� ��
A. ��H>0����S>0
B. ��H<0����S<0
C. ��H>0����S<0
D. ��H<0����S>0
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������й������˵������ȷ����
A.������̼���ŷ����γ��������Ҫԭ��
B.����ȼ�պ���Ļ�ʯȼ�����γ��������Ҫԭ��
C.�����pH��5.6
D.��������IJ����ɲ�ȡ�Ĵ�ʩ�Ƕ�ȼ�Ͻ�������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����и�������У������ۼ�������ǿ����������ȷ����( )
A��HF H2O NH3 CH4
B��CH4 NH3 H2O HF
C��H2O HF CH4 NH3
D��HF H2O CH4 NH3
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������й����ʵķ�����ȷ���ǣ�������
A. SO2��SiO2��CO������������
B. ϡ���������ᡢ�Ȼ�����Һ��Ϊ����
C. ���ᡢ���ᡢ������Ϊǿ��
D. ��ˮ��ˮ��������ˮ��Ϊ�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ڳ����£���0.1000mol/L������ζ�25mL0.100mol/Lna2CO3��Һ�����õζ���������ͼ��ʾ�����������Ũ�ȼ�Ĺ�ϵ����ȷ����

A. a��:c(CO32-)>c(Cl-)>c(HCO3-)
B. b��:c(Na+)+c(H+)= c(Cl-)+ c(HCO3-)+2c(CO32-)+c(OH-)
C. c��:c(OH-)+ c(CO32-)= c(H+)+c(H2CO3)
D. d��:c(Cl-)= c(Na+)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����������ȶ��Ļ�ѧ���ʣ����õ����¡��͵��¡���ǿ�ᡢ��ǿ���Լ���ǿ�ȡ����ܶȣ�������Ϊ��δ������������ս�Խ�������
(1)��̬��ԭ�Ӻ����_____���˶�״̬����ͬ�ĵ��ӣ��۵����Ų�ʽ____________________������ͬ���ڵ�Ԫ���У���̬ԭ�ӵ�δ�ɶԵ�����������ͬ����__�֡�
(2)�ѱȸ��ᡢ����Ӳ����һ�����˵Ľṹ���ϡ���Ӳ�ȱ������ԭ����___________________��
(3) TiCl4����������ȡ�ѵ��м���TiCl4���ӽṹ��CCl4��ͬ���ڳ����¶���Һ�塣TiCl4���ӵĿռ乹����_______________��Tiԭ�Ӳ�ȡ____�ӻ���ʽ��TiCl4�ȶ��Ա�CCl4�����ˮ�⣬�Դӽṹ������ԭ��_______________________________��
(4)���ѿ������һ�������������(CaTiO3)��ͬ����ṹ�IJ��ϣ��������صľ���ṹ�����߱��˺ܶ���ص��������ʣ����������ԡ�����Եȡ�CaTiO3����ṹ��ͼ��ʾ�����У�A����__________(д���ӷ���)��������λ����___________��
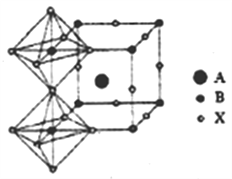
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���Ҷ����������ᣬ�����ǻ�ѧѧϰС���ͬѧ�Բ��ᾧ��(H2C2O4��xH2O)���е�̽����ѧϰ�Ĺ��̣�������벢Э������������ѧϰ������ͬѧ���о������ǣ�̽���ⶨ���ᾧ��(H2C2O4��xH2O)�е�xֵ��ͨ���������Ϻ������Ѱ����֪����������ˮ����ˮ��Һ����������KMnO4��Һ���еζ�����Ӧԭ��Ϊ2MnO4-+5H2C2O4+6H+=2Mn2++10CO2��+8H2O��
ѧϰС���ͬѧ��������µζ������ⶨxֵ��
����ȡ1.260g���ᾧ�壬�����Ƴ�100.00mLˮ��ҺΪ����Һ��
��ȡ25.00mL����Һ������ƿ�У��ټ���������ϡ���
����Ũ��Ϊ0.1000mol��L-1������KMnO4����Һ���еζ����ﵽ�ζ��յ�ʱ���ı���Һ10.00mL��
�ش��������⣺
��1���ζ�ʱ��������KMnO4��Һװ������ͼ�е�___________ (����������������)�ζ����С�

��2����ʵ�鵽��ζ��յ�ı�־��________________________________��
��3��ͨ���������ݣ����x=___________��
��4�����ζ����յ�ʱ���ӵζ��̶ܿ��߶��������ɴ˲�õ�xֵ��___________ (����ƫ������ƫС������������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ʵ��С���Ʊ�������أ�K2FeO4����̽�������ʡ�
���ϣ�K2FeO4Ϊ��ɫ���壬����KOH��Һ������ǿ�����ԣ������Ի�������Һ�п��ٲ���O2���ڼ�����Һ�н��ȶ���
��1���Ʊ�K2FeO4���г�װ���ԣ�
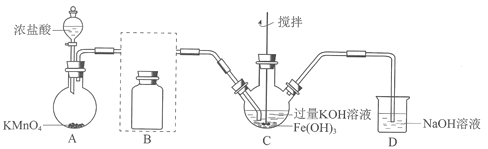
��AΪ��������װ�á�A�з�Ӧ����ʽ��________________���̱���ԭΪMn2+����
�ڽ�����װ��B�������������������Լ���_______
��C�еõ���ɫ�������Һ��C��Cl2�����ķ�Ӧ��
3Cl2+2Fe(OH)3+10KOH![]() 2K2FeO4+6KCl+8H2O�������________________��
2K2FeO4+6KCl+8H2O�������________________��
��2��̽��K2FeO4������
��ȡC����ɫ��Һ������ϡ���ᣬ��������ɫ���壬����Һa�������������к���Cl2��Ϊ֤���Ƿ�K2FeO4������Cl��������Cl2��������·�����
������ | ȡ����a���μ�KSCN��Һ����������Һ�ʺ�ɫ�� |
������ | ��KOH��Һ���ϴ��C�����ù��壬����KOH��Һ��K2FeO4�ܳ����õ���ɫ��Һb��ȡ����b���μ����ᣬ��Cl2������ |
i���ɷ���������Һ����֪a�к���______���ӣ��������ӵIJ��������ж�һ����K2FeO4��Cl����������������________________�������÷���ʽ��ʾ����
ii���������֤��K2FeO4������Cl������KOH��Һϴ�ӵ�Ŀ����________________��
�ڸ���K2FeO4���Ʊ�ʵ��ó���������Cl2________![]() �����������������������ʵ�������Cl2��
�����������������������ʵ�������Cl2��![]() ��������ǿ����ϵ�෴��ԭ����________________��
��������ǿ����ϵ�෴��ԭ����________________��
�����ϱ�����������Һ�е�������![]() ��
��![]() ����֤ʵ�����£�����Һb����MnSO4������H2SO4�Ļ����Һ�У�����Һ��dz��ɫ���������ܷ�֤��������
����֤ʵ�����£�����Һb����MnSO4������H2SO4�Ļ����Һ�У�����Һ��dz��ɫ���������ܷ�֤��������![]() ��
��![]() �����ܣ���˵�����ɣ������ܣ���һ�����ʵ�鷽����
�����ܣ���˵�����ɣ������ܣ���һ�����ʵ�鷽����
���ɻ���________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com