
| ||
| 6.15 |
| 10 |


 ���Ž�������С״Ԫϵ�д�
���Ž�������С״Ԫϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����

| 4100a |
| 22.4w |
| 4100a |
| 22.4w |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ��ɽ��ʡΫ���и����ڶ���ģ�⿼�����ۻ�ѧ�Ծ��������棩 ���ͣ�ʵ����
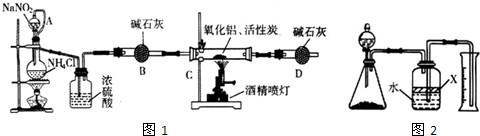
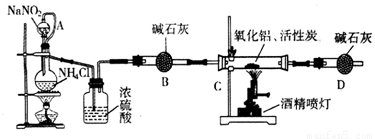
��������AIN����һ�����������ϣ��㷺Ӧ���ڼ��ɵ�·��������ij��ѧ�о�С�����õ������������ͻ���̿��ȡ�������������ͼʵ��װ�á�
�Իش�
��1��ʵ�����ñ���NaNO2�� NH4C��Һ��ȡ�����Ļ�ѧ����ʽΪ ��
��2��װ���з�Һ©����������ƿ֮��ĵ���A�������� ����д��ţ���
a.��ֹNaNO2 ������Һ���� b.��֤ʵ��װ�ò�©�� c.ʹNaNO2 ������Һ������
��3����ͼ���Ӻ�ʵ��װ�ã����װ�������Եķ����� ��
��4����ѧ�о�С���װ�ô����������⣬��˵���Ľ��İ취 ��
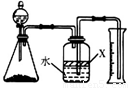
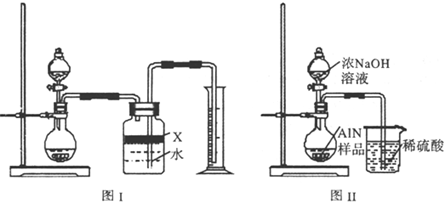
��5����Ӧ������ijͬѧ����ͼװ�ý���ʵ�����ⶨ��������Ʒ������������ʵ���е���������Բ��ƣ�����֪����������NaOH��Һ��Ӧ����Na[Al(OH)4]�Ͱ���
 ��
��
�ٹ��ƿ�е��Լ�X���ѡ�� ����д��ţ���
a.���� b.�ƾ� c.ֲ���� d.CCl4
�ڹ��ƿ�е�Һ��û��װ�����Ϸ����пռ䣩����ʵ����NH3������� ���ƫ����ƫС���������䡱����
����ʵ���г�ȡ��������Ʒ������Ϊ10.0g����ð��������Ϊ3.36L����״����������Ʒ��AlN����������Ϊ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ģ�� ���ͣ��ʴ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011������ʡ�����а�У�����߿���ѧģ���Ծ��������棩 ���ͣ������

�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com