
A��B��C��D��E���������о�����ͬһ�ַǽ���Ԫ�أ������ܷ�����ͼ��ʾ��ת����ϵ����Ԫ��(��R��ʾ)�ĵ�������NaOH��Һ��Ӧ�� ����(Na2RO3)��������
����(Na2RO3)��������
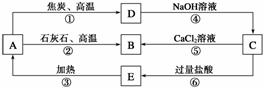
��ش��������⣺
(1)д�������ʵĻ�ѧʽ��A__________��B__________��C__________��D__________��E__________��
(2)д����Ӧ�ٵĻ�ѧ����ʽ��____________________________________________��
�÷�Ӧ���������뻹ԭ�������ʵ���֮��Ϊ________��
(3)д����Ӧ�ܵ����ӷ���ʽ��__________________________________________��
(4)д����Ӧ�ݵ����ӷ���ʽ��___________________________________________��
(5)H2CO3������ǿ��E�ģ��������ӷ���ʽ����֤����
________________________________________________________________________��
�𰸡�(1)SiO2��CaSiO3��Na2SiO3��Si��H2SiO3
(2)SiO2��2C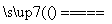 Si��2CO����1��2
Si��2CO����1��2
(3)Si��2OH����H2O===SiO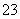 ��2H2��
��2H2��
(4)SiO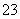 ��Ca2��===CaSiO3��
��Ca2��===CaSiO3��
(5)SiO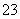 ��CO2��H2O===H2SiO3����CO
��CO2��H2O===H2SiO3����CO
�����������ġ�ͻ�ƿ�(1)���Ƿǽ���Ԫ��R�ĵ�������NaOH��Һ��Ӧ������(Na2RO3)��������˵���ǹ輰�仯����֮���ת������ͻ�ƿ�(2)���Ƿ�Ӧ���������¡���A�ڸ��������£����뽹̿��CaCO3������Ӧ����AΪSiO2���Ӷ�˳��������֪BΪCaSiO3��DΪSi��EΪH2SiO3��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�����仯�������ճ����������Ӧ�ù㷺���Ȼ���������ض��dz�����ˮ����������ͼΪ�Ʊ��Ȼ�������һ�������Ʊ�������صĹ������̡�
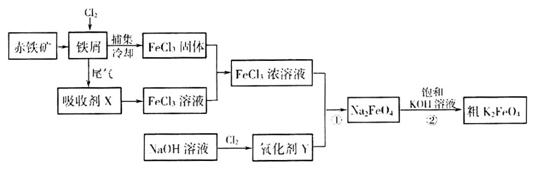 ��ش���������
��ش���������
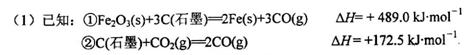
�ó�����Ϊԭ���ڸ�¯���������з�������Ҫ��ӦΪ
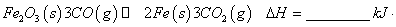 mol
mol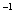 ��
��
(2)���ռ�x������Ϊ____________(д��ѧʽ)��
(3)������YΪ��84����Һ������Ч�ɷ֣����ڼ��������·�Ӧ�ٵ����ӷ���ʽΪ
__________________________________________________________________
(4)���̢�����ij�����½��еģ���Ӧ�Ļ�ѧ����ʽΪ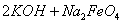 =
=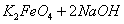 ��˵�����¶���
��˵�����¶���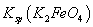 __________
__________ 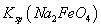 (�>����<��)��
(�>����<��)��
�ٶ��˹�����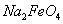 ��ȫת��Ϊ
��ȫת��Ϊ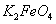 ���������Ƶôֲ�Ʒ
���������Ƶôֲ�Ʒ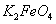 206.25t����Ʒ����Ϊ96������������������Ҫ������Y��������___________t��
206.25t����Ʒ����Ϊ96������������������Ҫ������Y��������___________t��
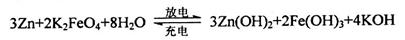 (5)���������һ�����Ͷ��ε�أ����ҺΪǿ����Һ�����ط�ӦΪ��
(5)���������һ�����Ͷ��ε�أ����ҺΪǿ����Һ�����ط�ӦΪ��
�ŵ�ʱ��صĸ�����ӦʽΪ______________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ͼ��ʾ����ע�����м�������Na2SO3���壬����������Ũ����(�Բ��Ӵ�ֽ��Ϊ)���������й�˵����ȷ����
A����ɫʯ����ֽ�ȱ�����ɫ
B��NaOH��Һ�����ڳ�ȥʵ���ж����SO2
C��ʪ��KI��������ֽδ������˵��SO2��������ǿ��I2
D��Ʒ����ֽ��մ��KMnO4��Һ����ֽ����ɫ��֤����SO2��Ư����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�±���Ԫ�����ڱ���һ���֡�
| �� | ��A | ��A | ��A | ��A | ��A | ��A | ��A |
| һ | �� | ||||||
| �� | �� | �� | �� | �� | �� | ||
| �� | �� | �� | �� | �� | |||
| �� | �� | �� | �� |
��1��Ԫ�� �ĵ��������³�Һ̬��Ԫ�� ���⻯�����ȶ���Ԫ�� ������������ˮ�����������ǿ��Ԫ�� �ĸ��������ˮ����ļ�����ǿ����д��Ԫ�ط��š�����
��2������Ԫ�آ�����γ��ȶ�������Ľṹʽ�� ��
��3������Ԫ�آ٢ۢ���ɵġ�A4B2C2�������ʣ������ں����������� ��
A�����Ӽ����ۼ� B����Ϊ���ۼ�
C����Ϊ���Թ��ۼ� D�����Թ��ۼ��ͷǼ��Թ��ۼ�
��4������Ԫ�� ���⻯��ķ��Ӽ�����������д��������š�����
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��������Һ����ʵ�飬�±��С������������롰��Һ����Ӧ��ϵ������� (����)
| ѡ�� | ���������� | ��Һ |
| A | ͨ��CO2����Һ����� | ����Na2CO3��Һ |
| B | ͨ��CO2����Һ����ǣ�����ͨCO2��������������ʧ | Na2SiO3��Һ |
| C | ͨ��CO2����Һ����ǣ��ټ���Ʒ����Һ����ɫ��ȥ | Ca(ClO)2��Һ |
| D | ͨ��CO2����Һ����ǣ�����ͨCO2��������������ʧ���ټ�������NaOH��Һ���ֱ���� | ����ʯ��ˮ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����������Ϣ��ҵ����Ҫ�Ļ������ϡ�ͨ����̼�ڸ����»�ԭ���������Ƶôֹ�(������������������)���ֹ���������Ӧ�������Ȼ���(��Ӧ�¶�450��500 ��)�����Ȼ��辭�ᴿ����������ԭ�ɵøߴ��衣������ʵ�����Ʊ����Ȼ����װ��ʾ��ͼ��
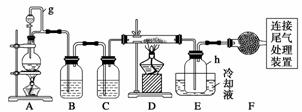
�����Ϣ���£�
a�����Ȼ�����ˮ����ˮ�⣻
b���������������ڸ����¾���������ֱ�ӷ�Ӧ������Ӧ���Ȼ��
c���й����ʵ������������±���
| ���� | SiCl4 | BCl3 | AlCl3 | FeCl3 | PCl5 |
| �е�/�� | 57.7 | 12.8 | �� | 315 | �� |
| �۵�/�� | ��70.0 | ��107.2 | �� | �� | �� |
| �����¶�/�� | �� | �� | 180 | 300 | 162 |
��ش��������⣺
(1)д��װ��A�з�����Ӧ�����ӷ���ʽ��_____________________________ ______
______
________________________________________________________________________��
(2)װ��A��g�ܵ�������____________��װ��C�е��Լ���__________��װ��E�е�hƿ��Ҫ��ȴ��������________________________________________________________
________________________________________________________________________��
(3)װ��E��hƿ�ռ����Ĵֲ����ͨ������(���ƶ������)�õ��ߴ������Ȼ��裬�����IJ������У�����Ԫ������ܻ����е�����Ԫ����________(��дԪ�ط���)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������CO2����ͨ��ˮ����(Na2SiO3��Һ)�У�Ȼ��������ɣ����ڸ����³�����գ����õ��Ĺ��������� (����)
A��Na2SiO3 B��Na2CO3��Na2SiO3
C��Na2CO3��SiO2 D��SiO2
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ǹ�ҵ���Ʊ�Na2S2O3�ķ���֮һ����Ӧԭ��Ϊ��
2Na2S+Na2CO3+4SO2==3Na2S2O3+CO2 ���÷�Ӧ��H>0��
ij�о�С����ʵ��������Ʊ�Na2S2O3��5H2O�������¡�
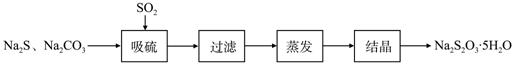
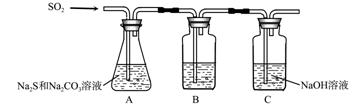 ��1������װ����ͼ��ʾ��
��1������װ����ͼ��ʾ��
��װ��B�������Ǽ���װ��A��SO2������Ч�ʣ�B���Լ��� ������SO2����Ч�ʵ͵�ʵ��������B����Һ ��
��Ϊ��ʹSO2������������ȫ���ڲ��ı�A����ҺŨ�ȡ�����������£����˼�ʱ���跴Ӧ���⣬���ɲ�ȡ�ĺ�����ʩ�� �� ����д��������
��2�����豾ʵ�����õ�Na2CO3������NaCl��NaOH�����ʵ�鷽�����м��顣������ʱCaCO3������Һ��pH=10.2��
��ѡ�Լ���������ϡ���ᡢAgNO3��Һ��CaCl2��Һ��Ca(NO3)2��Һ����̪��Һ������ˮ��pH�ơ��ձ����Թܡ��ι�
| ��� | ʵ����� | Ԥ������ | ���� |
| �� | ȡ������Ʒ���Թ��У�������������ˮ��������ܽ⣬___________________�� | _______________ | ��Ʒ��NaCl |
| �� | ��ȡ������Ʒ���ձ��У�������������ˮ����ֽ����ܽ⣬___________________�� | _______________ | ��Ʒ��NaOH |
��3��Na2S2O3��Һ�Ƕ���ʵ���еij����Լ����ⶨ��Ũ�ȵĹ������£�ȷ��ȡa g KIO3����ѧʽ����214�����������Һ���������KI�����H2SO4��Һ���μ�ָʾ������Na2S2O3��Һ�ζ����յ㣬����Na2S2O3��Һ�����ΪV mL����c(Na2S2O3)��_________mol��L��1����ֻ�г���ʽ���������㣩
��֪��Cr2O72����6I��+14H+=== 2Cr3+��3I2��7H2O 2S2O32����I2===S4O62����2I��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��ˮ�Ǿ����Դ���⣬�ں�ˮ�������ۺ����÷��棬�����λ��ȫ��ǰ�С��Ӻ�ˮ����ȡʳ�κ���Ĺ������£�

(1)���оٺ�ˮ���������ַ�����______________________��________________________��
(2)��NaCl��Һ���е�⣬�ڵ����п�ֱ�ӵõ��IJ�Ʒ��H2��________��________��H2��________��
(3)��������ѻ��Br2����������� ��Br2��ԭΪBr������Ŀ��Ϊ________________________________________________________________________��
��Br2��ԭΪBr������Ŀ��Ϊ________________________________________________________________________��
(4)�������SO2ˮ��Һ����Br2�������ʿɴ�95%���йط�Ӧ�����ӷ���ʽΪ________________________________________________________________________��
�ɴ˷�Ӧ��֪�������������⣬�ڹ�ҵ������Ӧ�������Ҫ������________________________________________________________________________��
(5)ij��ѧ�о���ѧϰС��Ϊ�˽�ӹ�ҵ�����ᴿ��ķ������������й����ϣ�Br2�ķе�Ϊ59 �棬����ˮ���ж��Ժ�ǿ��ʴ�ԡ����Dzι��������̺���������װ�ü�ͼ��

�������������ۣ�
��ͼ������B�����ƣ�________________��
������ʵ��װ�����������Ӿ��������������ܣ���ԭ��Ϊ________________________________________________________________________��
��ʵ��װ�����������ã�Ҫ�ﵽ�ᴿ���Ŀ�ģ���������ο��ƹؼ�������________________________________________________________________________��
��C��Һ�������ɫΪ________��Ϊ��ȥ�ò������Բ���������Cl2���������м���NaBr��Һ����ַ�Ӧ���ٽ��еķ��������___________________________________��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com