
| ”÷V |
| ”÷t |
| ”÷V |
| ”÷t |


 »īĮ¦æĪŹ±Ķ¬²½Į·Ļ°²įĻµĮŠ“š°ø
»īĮ¦æĪŹ±Ķ¬²½Į·Ļ°²įĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
£Ø¢ń£©£Ø6·Ö£©ĻÖÓŠĻĀĮŠĪļÖŹ£ŗ ¢ŁNaCl¾§Ģå””¢ŚŅŗĢ¬SO2””¢Ū“æ“×Ėį””¢ÜĮņĖį±µ””¢ŻĶ ¢Ž¾Ę¾«£ØC2H5OH£© ¢ßČŪ»ÆµÄKCl””¢ąNaOHČÜŅŗ””
ĒėÓĆŅŌÉĻĪļÖŹ»Ų“šĻĀĮŠĪŹĢā”££ØĢīŠņŗÅ£©
£Ø1£©ŌŚÉĻŹöדĢ¬ĻĀÄܵ¼µēµÄĪļÖŹŹĒ””””””””””””””””””””””””””””””””””””””””£»
£Ø2£©ŹōÓŚČõµē½āÖŹµÄŹĒ””””””””””””””””””””””””””””””””””£»
£Ø3£©ŹōÓŚ·Ēµē½āÖŹ£¬µ«ČÜÓŚĖ®ŗóµÄĖ®ČÜŅŗÄܵ¼µēµÄŹĒ””””””””””””””””””””””””£»
£Ø¢ņ£©£Ø4·Ö£©
ij»ÆѧŹµŃ銔×éĢ½¾æŹŠŹŪŹ³Óưד×ÖŠ“×ĖįµÄµÄ×¼Č·ÅØ¶Č£¬Č”25.00mLÄ³Ę·ÅĘŹ³ÓĆ°×
“×ӌ׶ŠĪĘæÖŠ£¬ŌŚŹµŃéŹŅÓĆÅضČĪŖcb mol/LµÄ±ź×¼NaOHČÜŅŗ¶ŌĘä½ųŠŠµĪ¶Ø”£
£Ø1£©×óĶ¼±ķŹ¾50mLµĪ¶Ø¹ÜÖŠŅŗĆęµÄĪ»ÖĆ£¬ČōAÓėCæĢ¶Č¼äĻą²īl mL£¬
A“¦µÄæĢ¶ČĪŖ25£¬µĪ¶Ø¹ÜÖŠŅŗĆę¶ĮŹżÓ¦ĪŖ””””””””””mL”£
£Ø2£©ĪŖĮĖ¼õŠ”ŹµŃéĪó²ī£¬øĆĶ¬Ń§Ņ»¹²½ųŠŠĮĖČż“ĪŹµŃ飬¼ŁÉčĆæ“Ī
ĖłČ”°×“×Ģå»ż¾łĪŖVmL£¬NaOH±ź×¼ŅŗÅضČĪŖc mo1/L£¬Čż“ĪŹµ
Ńé½į¹ū¼ĒĀ¼ČēĻĀ£ŗ
| ŹµŃé“ĪŹż | µŚŅ»“Ī | µŚ¶ž“Ī | µŚČż“Ī |
| ĻūŗÄNaOHČÜŅŗĢå»ż/mL | 26.02 | 25.35 | 25.30 |
“ÓÉĻ±ķæÉŅŌ擳ö£¬µŚŅ»“ĪŹµŃéÖŠ¼ĒĀ¼ĻūŗÄNaOHČÜŅŗµÄĢå»żĆ÷ĻŌ¶ąÓŚŗóĮ½“Ī£¬
ĘäŌŅņæÉÄÜŹĒ”””””””””””””””””£
A£®µĪ¶ØĒ°µĪ¶Ø¹Ü¼ā×ģÓŠĘųÅŻ£¬µĪ¶Ø½įŹųĪŽĘųÅŻ
B£®Ź¢×°±ź×¼ŅŗµÄµĪ¶Ø¹Ü×°ŅŗĒ°ÓĆÕōĮóĖ®ČóĻ“¹ż£¬Ī“ÓƱź×¼ŅŗČóĻ“
C£®µŚŅ»“ĪµĪ¶ØÓƵÄ׶ŠĪĘæĪ“ČóĻ“
D£®µĪ¶Ø½įŹųŹ±£¬ø©ŹÓ¶ĮŹż
£Ø3£©øł¾ŻĖłøųŹż¾Ż£¬Š“³ö¼ĘĖćøưד×ÖŠ“×ĖįµÄĪļÖŹµÄĮæÅØ¶ČµÄ±ķ“ļŹ½(²»±Ų»Æ¼ņ)£ŗ
c£½”””””””””” ”””””” ”£
£Ø¢ó£©£Ø15·Ö£©
ŅŃÖŖ£ŗŌŚ25Ź±H2OH++OH-””KW=10-14”” CH3COOH
H++ CH3COO£””Ka=1.8”Į10-5
£Ø1£©Č”ŹŹĮæ“×ĖįČÜŅŗ£¬¼ÓČėÉŁĮæ“×ĖįÄĘ¹ĢĢ壬“ĖŹ±ČÜŅŗÖŠC£ØH+£©ÓėC£ØCH3COOH£©
µÄ±ČÖµ”” ””£ØĢī”°Ōö“ó”±»ņ”°¼õŠ””±»ņ”°²»±ä”±£©
£Ø2£©“×ĖįÄĘĖ®½āµÄĄė×Ó·½³ĢŹ½ĪŖ”””””””””””””””””£µ±ÉżøßĪĀ¶ČŹ±£¬C(OH”Ŗ)½«””””””””
£ØĢī”°Ōö“ó”±”°¼õŠ””±”°²»±ä”±£©£»
£Ø3£©0£®5mol”¤L-1“×ĖįÄĘČÜŅŗpHĪŖm£¬ĘäĖ®½āµÄ³Ģ¶Č£ØŅŃĖ®½āµÄ“×ĖįÄĘÓėŌÓŠ“×ĖįÄĘ
µÄ±ČÖµ£©ĪŖa£»1mol”¤L-1“×ĖįÄĘČÜŅŗpHĪŖn£¬Ė®½āµÄ³Ģ¶ČĪŖb£¬ŌņmÓėnµÄ¹ŲĻµ
ĪŖ”””””””””””””””””” £¬aÓėbµÄ¹ŲĻµĪŖ””””””””””£ØĢī”°“óÓŚ”±”°Š”ÓŚ”±”°µČÓŚ”±£©£»
£Ø4£©½«µČĢå»żµČÅØ¶ČµÄ“×ĖįŗĶĒāŃõ»ÆÄĘČÜŅŗ»ģŗĻŗó£¬ĖłµĆČÜŅŗÖŠĄė×ÓÅضČÓɓ󵽊”µÄĖ³ŠņŹĒ”””””””””””””””” ”””””” ”£
£Ø5£©Čō“×ĖįŗĶĒāŃõ»ÆÄĘČÜŅŗ»ģŗĻŗópH<7£¬Ōņc£ØNa+£©_______________ c£ØCH3COO££©£ØĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©£¬
£Ø6£©ČōÓÉpH£½3µÄHAČÜŅŗV1mLÓėpH£½11µÄNaOH{ČÜŅŗV2 mL”£»ģŗĻ¶ųµĆ£¬ŌņĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ____________”£
A£®Čō·“Ó¦ŗóČÜŅŗ³ŹÖŠŠŌ£¬Ōņc£ØH+£©+c£ØOH££©£½2”Į10£7mol”¤L£1
B£®ČōV1=V2£¬·“Ó¦ŗóČÜŅŗpHŅ»¶ØµČÓŚ7
C£®Čō·“Ó¦ŗóČÜŅŗ³ŹĖįŠŌ£¬ŌņV1Ņ»¶Ø“óÓŚV2
D£®Čō·“Ó¦ŗóČÜŅŗ³Ź¼īŠŌ£¬ŌņV1Ņ»¶ØŠ”ÓŚV2
£Ø7£©ŌŚÄ³ČÜŅŗÖŠŗ¬Mg2+”¢Cd2+”¢Zn2+ČżÖÖĄė×ÓµÄÅØ¶Č¾łĪŖ0.01mol”¤L-1”£ĻņĘäÖŠ¼ÓČė¹Ģ
Ģå“×ĖįÄĘŗó²āµĆČÜŅŗµÄC(OH-)ĪŖ2.2”Į10-5mol”¤L-1£¬ŅŌÉĻČżÖÖ½šŹōĄė×ÓÖŠ
”””””””” ÄÜÉś³É³Įµķ£¬ŌŅņŹĒ”””” ””
£ØKSP£ŪMg£ØOH£©2£Ż=1.8”Į10-11”¢KSP£ŪZn£ØOH£©2£Ż=1.2”Į10-17”¢KSP£ŪCd£ØOH£©2£Ż=2.5”Į10-14£©
£Ø8£©Č”10mL0.5mol”¤L-1ŃĪĖįČÜŅŗ£¬¼ÓĖ®Ļ”ŹĶµ½500mL£¬ŌņøĆČÜŅŗÖŠÓÉĖ®µēĄė³öµÄc£ØH+£©
=”””””” ”””£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2012-2013Ń§ÄźÉ½¶«Ź”¼ĆÄĻŹŠøßČż4ŌĀ¹®¹ĢŠŌѵĮ·Ąķ×Ū»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗŹµŃéĢā
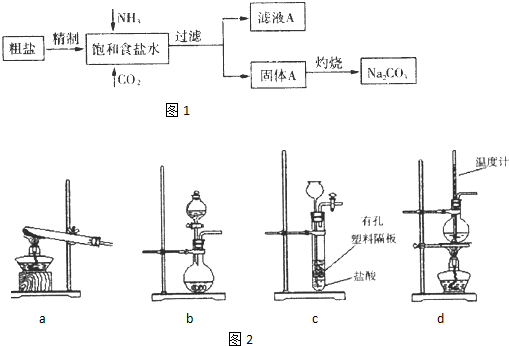
“æ¼ī(Na2CO3)ŌŚÉś²śÉś»īÖŠ¾ßÓŠ¹ć·ŗµÄÓĆĶ¾”£ŅŌĻĀŹĒŹµŃéŹŅÄ£ÄāÖĘ¼īŌĄķÖĘČ”Na2CO3µÄĮ÷³ĢĶ¼

ŅŃÖŖ:Ļņ±„ŗĶŹ³ŃĪĖ®ÖŠĶØČėNH3£¬CO2ŗó·¢ÉśŗĶ·“Ó¦ĪŖNaCl£«NH3£«CO2£«H2O NaHCO3”ż£«NH4Cl,Ēė»Ų“šŅŌĻĀĪŹĢā£ŗ
NaHCO3”ż£«NH4Cl,Ēė»Ų“šŅŌĻĀĪŹĢā£ŗ
£Ø1£©“ÖŃĪÖŠŗ¬ÓŠµÄŌÓÖŹĄė×ÓÓŠCa2+£¬Mg2+£¬SO42-µČ”£
¾«ÖĘ³żŌӵIJ½ÖčĖ³Šņa”ś_______”ś________”ś________”śb(Ģī×ÖÄø±ąŗÅ£©”£
a£®“ÖŃĪČܽā£¬ĀĖČ„³ĮŌü£®b£®¼ÓČėŃĪĖįµ÷pH£»c£®¼ÓČėBa(OH)2ČÜŅŗ£»d£®¼ÓČėNa2CO3ČÜŅŗ£»e£®¹żĀĖ
Ļņ±„ŗĶŹ³ŃĪĖ®ÖŠĻČĶØČėNH3£¬ŗóĶØČĖCO2£¬ĄķÓÉŹĒ_____________________”£
£Ø2£©×ĘÉÕ¹ĢĢåAÖĘNa2CO3ŌŚ_____Ģī×ÖÄøŠņŗÅ£©ÖŠ½ųŠŠ”£
a£®ŪįŪö b£®Õō·¢Ćó c£®ÉÕ± d£®×¶ŠĪĘæ
Ö¤Ć÷ĀĖŅŗAÖŠŗ¬ÓŠNH4+µÄ·½·ØŹĒ__________________________________________________________”£
¶ŌĀĖŅŗA½ųŠŠÖŲ½į¾§Äܹ»»ńµĆNH4HCO3£¬ĻņpH=13ŗ¬Na+£¬K+µÄČÜŅŗÖŠ¼ÓČėÉŁĮæNH4HCO3”£Ź¹pH½µµĶ£¬·“Ó¦µÄĄė×Ó·½³ĢŹ½____________________________________”£
£Ø3£©ĻĀĶ¼×°ÖĆÖŠ³£ÓĆÓŚŹµŃéŹŅÖʱøCO2µÄŹĒ_____(Ģī×ÖÄø±ąŗÅ£©£»ÓĆbŹ¾ŅāµÄ×°ÖĆÖʱøNH3£¬·ÖŅŗĀ©¶·ÖŠŹ¢·ÅµÄŹŌ¼Į______(ĢīŹŌ¼ĮĆū³Ę£©£¬ÉÕĘæÄŚæɼÓČėµÄ¹ĢĢåŹŌ¼Į__________£ØĢīŹŌ¼ĮĆū³Ę£©”£

£Ø4£©Ņ»ÖÖĢģČ»¼ī¾§Ģå³É·ÖŹĒaNa2CO3”¤bNa2CO3”¤cH2O£¬Ä³Ķ¬Ń§ĄūÓĆĻĀĮŠĢį¹©µÄŹŌ¼Į£¬Éč¼ĘĮĖČēĻĀ¼ņµ„ŗĻĄķ²ā¶ØNa2CO3µÄÖŹĮæ·ÖŹżµÄŹµæ„·½°ø”££ØŅĒĘ÷×ŌŃ”£©Ēė°ŃŹµŃé·½°øĢīČ«£ŗ¹©Ń”ŌńµÄŹŌ¼Į£ŗ1.0mol/LH2SO4ČÜŅŗ”¢1.0mol/L BaCl2ČÜŅŗ”¢Ļ”°±Ė®”¢¼īŹÆ»Ņ”¢Ca(OH)2ČÜŅŗ”¢ÕōĮóĖ®
¢Ł³ĘČ”m1gŅ»¶ØĮæĢģČ»¼ī¾§Ģåѳʷ£¬ČÜÓŚŹŹĮæÕōĮóĖ®ÖŠ”£
¢Ś_________________________________________________________________”£
¢Ū_________________________________________________________________”£
¢Ü¼ĘĖćĢģČ»¼ī¾§ĢåÖŠŗ¬Na2CO3µÄÖŹĮæ·ÖŹż”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013½ģø£½ØŹ¦“óø½ÖŠø߶žÉĻŃ§ĘŚĘŚÄ©æ¼ŹŌ»ÆѧŹŌ¾ķ ĢāŠĶ£ŗĢīæÕĢā
£Ø¢ń£©£Ø6·Ö£©ĻÖÓŠĻĀĮŠĪļÖŹ£ŗ ¢ŁNaCl¾§Ģå””¢ŚŅŗĢ¬SO2””¢Ū“æ“×Ėį””¢ÜĮņĖį±µ””¢ŻĶ ¢Ž¾Ę¾«£ØC2H5OH£© ¢ßČŪ»ÆµÄKCl””¢ąNaOHČÜŅŗ””
ĒėÓĆŅŌÉĻĪļÖŹ»Ų“šĻĀĮŠĪŹĢā”££ØĢīŠņŗÅ£©
£Ø1£©ŌŚÉĻŹöדĢ¬ĻĀÄܵ¼µēµÄĪļÖŹŹĒ”””””””””””””””””””””””””””””””””””””””” £»
£Ø2£©ŹōÓŚČõµē½āÖŹµÄŹĒ”””””””””””””””””””””””””””””””””” £»
£Ø3£©ŹōÓŚ·Ēµē½āÖŹ£¬µ«ČÜÓŚĖ®ŗóµÄĖ®ČÜŅŗÄܵ¼µēµÄŹĒ”””””””””””””””””””””””” £»
£Ø¢ņ£©£Ø4·Ö£©
ij»ÆѧŹµŃ銔×éĢ½¾æŹŠŹŪŹ³Óưד×ÖŠ“×ĖįµÄµÄ×¼Č·ÅØ¶Č£¬Č”25.00mLÄ³Ę·ÅĘŹ³ÓĆ°×
“×ӌ׶ŠĪĘæÖŠ£¬ŌŚŹµŃéŹŅÓĆÅضČĪŖcb mol/LµÄ±ź×¼NaOHČÜŅŗ¶ŌĘä½ųŠŠµĪ¶Ø”£
£Ø1£©×óĶ¼±ķŹ¾50mLµĪ¶Ø¹ÜÖŠŅŗĆęµÄĪ»ÖĆ£¬ČōAÓėCæĢ¶Č¼äĻą²īl mL£¬
A“¦µÄæĢ¶ČĪŖ25£¬µĪ¶Ø¹ÜÖŠŅŗĆę¶ĮŹżÓ¦ĪŖ””””””””””mL”£

£Ø2£©ĪŖĮĖ¼õŠ”ŹµŃéĪó²ī£¬øĆĶ¬Ń§Ņ»¹²½ųŠŠĮĖČż“ĪŹµŃ飬¼ŁÉčĆæ“Ī
ĖłČ”°×“×Ģå»ż¾łĪŖVmL£¬NaOH±ź×¼ŅŗÅضČĪŖc mo1/L£¬Čż“ĪŹµ
Ńé½į¹ū¼ĒĀ¼ČēĻĀ£ŗ
|
ŹµŃé“ĪŹż |
µŚŅ»“Ī |
µŚ¶ž“Ī |
µŚČż“Ī |
|
ĻūŗÄNaOHČÜŅŗĢå»ż/mL |
26.02 |
25.35 |
25.30 |
“ÓÉĻ±ķæÉŅŌ擳ö£¬µŚŅ»“ĪŹµŃéÖŠ¼ĒĀ¼ĻūŗÄNaOHČÜŅŗµÄĢå»żĆ÷ĻŌ¶ąÓŚŗóĮ½“Ī£¬
ĘäŌŅņæÉÄÜŹĒ”””””””””””””””” ”£
A£®µĪ¶ØĒ°µĪ¶Ø¹Ü¼ā×ģÓŠĘųÅŻ£¬µĪ¶Ø½įŹųĪŽĘųÅŻ
B£®Ź¢×°±ź×¼ŅŗµÄµĪ¶Ø¹Ü×°ŅŗĒ°ÓĆÕōĮóĖ®ČóĻ“¹ż£¬Ī“ÓƱź×¼ŅŗČóĻ“
C£®µŚŅ»“ĪµĪ¶ØÓƵÄ׶ŠĪĘæĪ“ČóĻ“
D£®µĪ¶Ø½įŹųŹ±£¬ø©ŹÓ¶ĮŹż
£Ø3£©øł¾ŻĖłøųŹż¾Ż£¬Š“³ö¼ĘĖćøưד×ÖŠ“×ĖįµÄĪļÖŹµÄĮæÅØ¶ČµÄ±ķ“ļŹ½(²»±Ų»Æ¼ņ)£ŗ
c£½”””””””””” ”””””” ”£
£Ø¢ó£©£Ø15·Ö£©
ŅŃÖŖ£ŗŌŚ25Ź±H2O H++OH-””KW=10-14”” CH3COOH
H++OH-””KW=10-14”” CH3COOH
 H++ CH3COO£””Ka=1.8”Į10-5
H++ CH3COO£””Ka=1.8”Į10-5
£Ø1£©Č”ŹŹĮæ“×ĖįČÜŅŗ£¬¼ÓČėÉŁĮæ“×ĖįÄĘ¹ĢĢ壬“ĖŹ±ČÜŅŗÖŠC£ØH+£©ÓėC£ØCH3COOH£©
µÄ±ČÖµ”” ”” £ØĢī”°Ōö“ó”±»ņ”°¼õŠ””±»ņ”°²»±ä”±£©
£Ø2£©“×ĖįÄĘĖ®½āµÄĄė×Ó·½³ĢŹ½ĪŖ”””””””””””””””” ”£µ±ÉżøßĪĀ¶ČŹ±£¬C(OH”Ŗ)½«””””””””
£ØĢī”°Ōö“ó”±”°¼õŠ””±”°²»±ä”±£©£»
£Ø3£©0£®5mol”¤L-1“×ĖįÄĘČÜŅŗpHĪŖm£¬ĘäĖ®½āµÄ³Ģ¶Č£ØŅŃĖ®½āµÄ“×ĖįÄĘÓėŌÓŠ“×ĖįÄĘ
µÄ±ČÖµ£©ĪŖa£»1mol”¤L-1“×ĖįÄĘČÜŅŗpHĪŖn£¬Ė®½āµÄ³Ģ¶ČĪŖb£¬ŌņmÓėnµÄ¹ŲĻµ
ĪŖ”””””””””””””””””” £¬aÓėbµÄ¹ŲĻµĪŖ””””””””””£ØĢī”°“óÓŚ”±”°Š”ÓŚ”±”°µČÓŚ”±£©£»
£Ø4£©½«µČĢå»żµČÅØ¶ČµÄ“×ĖįŗĶĒāŃõ»ÆÄĘČÜŅŗ»ģŗĻŗó£¬ĖłµĆČÜŅŗÖŠĄė×ÓÅضČÓɓ󵽊”µÄĖ³ŠņŹĒ”””””””””””””””” ”””””” ”£
£Ø5£©Čō“×ĖįŗĶĒāŃõ»ÆÄĘČÜŅŗ»ģŗĻŗópH<7£¬Ōņc£ØNa+£©_______________ c£ØCH3COO££©£ØĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©£¬
£Ø6£©ČōÓÉpH£½3µÄHAČÜŅŗV1mLÓėpH£½11µÄNaOH{ČÜŅŗV2 mL”£»ģŗĻ¶ųµĆ£¬ŌņĻĀĮŠĖµ·Ø²»ÕżČ·µÄŹĒ____________”£
A£®Čō·“Ó¦ŗóČÜŅŗ³ŹÖŠŠŌ£¬Ōņc£ØH+£©+c£ØOH££©£½2”Į10£7mol”¤L£1
B£®ČōV1=V2£¬·“Ó¦ŗóČÜŅŗpHŅ»¶ØµČÓŚ7
C£®Čō·“Ó¦ŗóČÜŅŗ³ŹĖįŠŌ£¬ŌņV1Ņ»¶Ø“óÓŚV2
D£®Čō·“Ó¦ŗóČÜŅŗ³Ź¼īŠŌ£¬ŌņV1Ņ»¶ØŠ”ÓŚV2
£Ø7£©ŌŚÄ³ČÜŅŗÖŠŗ¬Mg2+”¢Cd2+”¢Zn2+ČżÖÖĄė×ÓµÄÅØ¶Č¾łĪŖ0.01mol”¤L-1”£ĻņĘäÖŠ¼ÓČė¹Ģ
Ģå“×ĖįÄĘŗó²āµĆČÜŅŗµÄC(OH-)ĪŖ2.2”Į10-5mol”¤L-1£¬ŅŌÉĻČżÖÖ½šŹōĄė×ÓÖŠ
”””””””” ÄÜÉś³É³Įµķ£¬ŌŅņŹĒ”””” ””
£ØKSP£ŪMg£ØOH£©2£Ż=1.8”Į10-11”¢KSP£ŪZn£ØOH£©2£Ż=1.2”Į10-17”¢KSP£ŪCd£ØOH£©2£Ż=2.5”Į10-14£©
£Ø8£©Č”10mL0.5mol”¤L-1ŃĪĖįČÜŅŗ£¬¼ÓĖ®Ļ”ŹĶµ½500mL£¬ŌņøĆČÜŅŗÖŠÓÉĖ®µēĄė³öµÄc£ØH+£©
=”””””” ”””£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
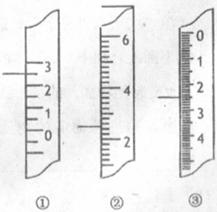 (4·Ö)£®(1)ĪĀ¶Č¼Ę³£ÓĆĄ“¼ą²ā»ņæŲÖĘ»ÆѧŹµŃéµÄĪĀ¶Č”£
(4·Ö)£®(1)ĪĀ¶Č¼Ę³£ÓĆĄ“¼ą²ā»ņæŲÖĘ»ÆѧŹµŃéµÄĪĀ¶Č”£
ĒėĖµĆ÷ĻĀĮŠŹµŃéÖŠĪĀ¶Č¼ĘµÄĖ®ŅųĒņĖł“¦µÄĪ»ÖĆ£ŗŹµŃéŹŅÖĘŅŅĻ©____________________£¬ÓĆÕōĮó·Ø½ųŠŠ»ģŗĻĪļµÄ·ÖĄė_______________________________”£
(2)ÓŅĶ¼·Ö±šŹĒĪĀ¶Č¼Ę”¢ĮæĶ²”¢µĪ¶Ø¹ÜµÄŅ»²æ·Ö£¬
ĻĀŹö¶ĮŹż(×ó²ąŹµĻß±ź×¢Ī»ÖĆ)¼°Ėµ·ØÕżČ·µÄŹĒ_______ ”£
A£®¢ŁŹĒĮæĶ²£¬¶ĮŹżĪŖ2.5mL
B£®¢ŚŹĒĮæĶ²£¬¶ĮŹżĪŖ2.5 mL
 C£®¢ŪŹĒµĪ¶Ø¹Ü£¬¶ĮŹżĪŖ3.5 mL
C£®¢ŪŹĒµĪ¶Ø¹Ü£¬¶ĮŹżĪŖ3.5 mL
D£®¢ŁŹĒĪĀ¶Č¼Ę£¬¶ĮŹżĪŖ2.
(3)ijĶ¬Ń§ÓĆÓŅĶ¼ĖłŹ¾×°ÖĆ£¬½«ĘųĢå·¢Éś×°ÖĆÖŠ²śÉśµÄĀČ»ÆĒāÖ±½ÓĶØČėĖ®ÖŠÅäÖĘÉŁĮæŃĪĖį£¬½į¹ū·¢ÉśĮĖĖ®µÄµ¹Īü”£ŌŚ²»Ōö¼ÓŅĒĘ÷(æÉŃ”Ōń±ŲŠčµÄŹŌ¼Į)µÄĒ°ĢįĻĀ½«ÓŅĶ¼×°ÖĆÉŌ¼ÓøĽų£¬¼“æÉ°²Č«µŲŌŚŹŌ¹ÜÖŠÓĆĖ®ĪüŹÕĀČ»ÆĒā£¬øĽųµÄ·½·ØŹĒ______________
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com