
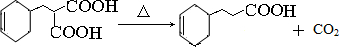
��19�֣� ij����A����ͼ��ʾ�Ĺ���ת��Ϊ������D����֪DΪǿ�ᣬ��ش��������⣺
��1����A������Ϊ����ɫ���嵥�ʣ�B���д̼�����ζ����ɫ���塣
�����A��Ԫ�������ڱ�λ��Ϊ������������������������
��Ӧ���ӵĽṹʾ��ͼΪ����������������������������������
��д��B��C��Ӧ�Ļ�ѧ����ʽ������������������������������������
������Bͨ��Ʒ����Һ�У���������������������������������B��
�ԣ�����Bͨ����ɫ��ʯ����Һ�У���������������������������������������
������B��ˮ��Һ�� �ԡ�
��D��Ũ��Һ�ڼ��������¿���ͭ��Ӧ������B���壬�÷�Ӧ�Ļ�ѧ����ʽΪ��
��������������������������������������
��2����A�ڳ�����Ϊ���壬����ʹʪ��ĺ�ɫʯ����ֽ������C�Ǻ���ɫ���塣
��A��C�Ļ�ѧʽ�ֱ��ǣ�A����������������C
��д��A��B��Ӧ�Ļ�ѧ����ʽ��������������������������������
������Aͨ����з�̪��ˮ�У������������� ���÷���ʽ��ʾ��ԭ��
��
��D��Ũ��Һ����ͭ��Ӧ������C���壬��÷�Ӧ�Ļ�ѧ����ʽΪ��
��������������������������������������������������
(��19��)��2���١�����������VIA�塡�������� (��1��)
�ڡ�2SO2+O22SO3 (2��)
�����ۡ�Ʒ����Һ��ɫ�� Ư�ף���Һ��죻 ��(��1��)
�ܡ�Cu+2 H2SO4��Ũ����CuSO4 +SO2��+2H2O (2��)
����2���١�NH3�� NO2 (��1��) ��4NH3+5O2 == 4NO+6H2O(2��)
��������Һ���(1��)����NH3+H2ONH3�� H2ONH4++OH- (2��)
��Cu��4HNO3(Ũ)��Cu(NO3)2��2NO2����2H2O (2��)
����:



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
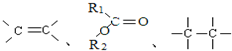 ��ԭ�Ӻͻ��ţ��ش������й����⣺
��ԭ�Ӻͻ��ţ��ش������й����⣺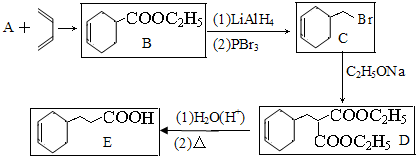
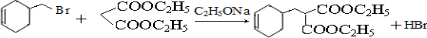
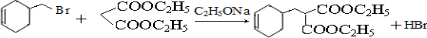

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
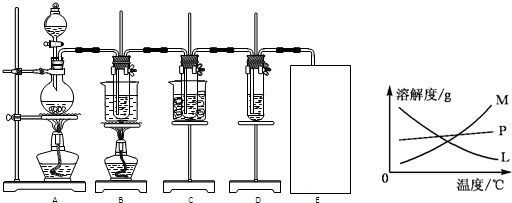
| ||
| ||
| ||
| ʵ������ | ԭ�� |
| ��Һ�������ɫ��Ϊ �� �� ɫ |
������ˮ��Ӧ���ɵ�H+ʹʯ���ɫ |
| �����Һ��Ϊ��ɫ | ������ˮ��Ӧ���ɵ�HClO��ʯ������Ϊ��ɫ���� ������ˮ��Ӧ���ɵ�HClO��ʯ������Ϊ��ɫ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2010-2011ѧ�긣��ʡ�����а��أ��У���һ��ѧ������������ѧ�Ծ� ���ͣ�ʵ����
��19�֣�ij����A����ͼ��ʾ�Ĺ���ת��Ϊ������D����֪DΪǿ�ᣬ��ش��������⣺
��1����A������Ϊ����ɫ���嵥�ʣ�B���д̼�����ζ����ɫ���塣
�����A��Ԫ�������ڱ�λ��Ϊ������������������������
��Ӧ���ӵĽṹʾ��ͼΪ����������������������������������
��д��B��C��Ӧ�Ļ�ѧ����ʽ������������������������������������
������Bͨ��Ʒ����Һ�У���������������������������������B��
�ԣ�����Bͨ����ɫ��ʯ����Һ�У���������������������������������������
������B��ˮ��Һ�� �ԡ�
��D��Ũ��Һ�ڼ��������¿���ͭ��Ӧ������B���壬�÷�Ӧ�Ļ�ѧ����ʽΪ��
��������������������������������������
��2����A�ڳ�����Ϊ���壬����ʹʪ��ĺ�ɫʯ����ֽ������C�Ǻ���ɫ���塣
��A��C�Ļ�ѧʽ�ֱ��ǣ�A����������������C
��д��A��B��Ӧ�Ļ�ѧ����ʽ��������������������������������
������Aͨ����з�̪��ˮ�У������������� ���� ����ʽ��ʾ��ԭ��
����ʽ��ʾ��ԭ��
��
��D��Ũ��Һ����ͭ��Ӧ������C���壬��÷�Ӧ�Ļ�ѧ����ʽΪ��
��������������������������������������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�츣��ʡ�����а��أ��У���һ��ѧ������������ѧ�Ծ� ���ͣ�ʵ����
��19�֣� ij����A����ͼ��ʾ�Ĺ���ת��Ϊ������D����֪DΪǿ�ᣬ��ش��������⣺

��1����A������Ϊ����ɫ���嵥�ʣ�B���д̼�����ζ����ɫ���塣
�����A��Ԫ�������ڱ�λ��Ϊ������������������������
��Ӧ���ӵĽṹʾ��ͼΪ����������������������������������
��д��B��C��Ӧ�Ļ�ѧ����ʽ������������������������������������
������Bͨ��Ʒ����Һ�У���������������������������������B��
�ԣ�����Bͨ����ɫ��ʯ����Һ�У���������������������������������������
������B��ˮ��Һ�� �ԡ�
��D��Ũ��Һ�ڼ��������¿���ͭ��Ӧ������B���壬�÷�Ӧ�Ļ�ѧ����ʽΪ��
��������������������������������������
��2����A�ڳ�����Ϊ���壬����ʹʪ��ĺ�ɫʯ����ֽ������C�Ǻ���ɫ���塣
��A��C�Ļ�ѧʽ�ֱ��ǣ�A����������������C
��д��A��B��Ӧ�Ļ�ѧ����ʽ��������������������������������
������Aͨ����з�̪��ˮ�У������������� ���÷���ʽ��ʾ��ԭ��
��
��D��Ũ��Һ����ͭ��Ӧ������C���壬��÷�Ӧ�Ļ�ѧ����ʽΪ��
��������������������������������������������������
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com