
��9�֣�ij�о���ѧϰС�����о���������Ư������ʱ���ӡ�������Ư������ʵ������������ˮ��Ӧ���ɵĴ������Ư�����á��õ�������Ϊ��̽�����������Ư�����õ����Ƕ������������Ƕ���������ˮ���õIJ����С�����������ʵ�顣��ش�������⡣
��1��Ϊ��̽��SO2�ܷ�ʹƷ����ɫ����ͬѧѡ������ȷ��ҩƷ�����������ͼ��ʾʵ��װ�ã���ָ��ʵ��װ��ͼ����еIJ�����֮����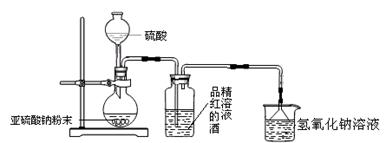
�� ��
�� ��
��2����ͬѧѡ������ȷװ�ú�ʵ���п��ƶ��������Դ�Լÿ��3�����ݵ��ٶ�ͨ��Ʒ��ľƾ���Һʱ������һСʱ��Ʒ���Բ���ɫ��Ϊ�ˣ�����ΪʹƷ���ˮ��Һ��ɫ���������� ��
��3����ͬѧ��һ��ʵ�����£�ȡ������ͬŨ�ȵ�Ʒ��ˮ��Һ����֧�Թ��У��ٷֱ���������������ƹ�������������ƹ��壬��֧�Թ��е�Ʒ�춼��ɫ�����ó����ۣ�ʹƷ����ɫ�����϶���HSO3-��SO32-������Ϊ���Ľ����Ƿ���ȷ �� �������� ��
��4����������װ��̽��SO2��ijЩ��ѧ���ʡ�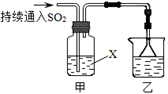
��װ���ҵ������� ��
����XΪNa2S��Һ���۲쵽��Һ�г��ֵ���ɫ���ǣ�˵��SO2���� ��
a�������� b����ԭ��
c��Ư���� d.���ȶ���
�����Լ�XΪCa(ClO)2��Һ���ɹ۲쵽��ɫ�������ɣ���ɸù��̵����ӷ���ʽ�� Ca2��+
Ca2��+ ClO��+
ClO��+ SO2+
SO2+ H2O��
H2O�� ��+
��+ Cl��+
Cl��+ SO42��+
SO42��+ ��
��
��1����ȱ�ٶ�������ĸ���װ�ã�1�֣��� �ڲ���ʹ�ó���©����1�֣�
��2�� H2SO3��HSO3-��SO32����1�֣�
��3������ȷ��1�֣� ��Ϊ����������Ӻ�������������Ӷ���ˮ�����������ᣨ1�֣���
��4����β����������1�֣���a����1�֣�
��Ca2++2ClO-+2SO2+2H2O=CaSO4��+2Cl-+SO42-+4H+����2�֣�
���������������1����ͬѧ��ʵ��Ŀ����Ϊ��̽�����������Ư�����õ����Ƕ������������Ƕ���������ˮ���õIJ�����Զ�����������Ǹ���ģ�ȱ�ٸ���װ�ã���β����ó���©��ʢ�����ᣬӦ���÷�Һ©����
��2����������ͨ��Ʒ��ľƾ���Һ����Һ����ɫ����ͨ��Ʒ���ˮ��Һ��ɫ����������ͨ��ƾ���Һ����������ˮ��Һ�в�ͬ��������Ҫ��H2SO3��HSO3-��SO32������Щ���Ӷ�������ʹƷ����ɫ�����ӣ�
��3���������ƹ�������������ƹ�������ˮ��ˮ�ⶼ�õ�H2SO3�����Բ����ж���ʹƷ����ɫ�����϶���HSO3-��SO32-����ͬѧ���ж��Dz���ȷ�ģ�
��4����װ������������ʣ��Ķ�������ֹ��Ⱦ������
��Na2S���������Ӧ����S���ʣ����������е�SԪ�صĻ��ϼ۽��ͣ�˵������������������ԣ���ѡa��
��Ca(ClO)2��Һ�����������������ԭ��Ӧ��ʣ��÷���ʽ�еij�����CaSO4������Ԫ���غ㣬�ж���һ������H+��SO2��SԪ�ػ��ϼ�����2�ۣ�Ca(ClO)2��ClԪ�����彵��2�ۣ����Է���ʽ��SO2��Ca(ClO)2��ϵ������1�����ݵ���غ�۲취��ƽ�������ʵ�ϵ�������ӷ���ʽΪCa2++2ClO-+2SO2+2H2O=CaSO4��+2Cl-+SO42-+4H+��
���㣺��������������ȡʵ�顢���ʵ���֤ʵ�飬�Է�Ӧ�ķ��������ӷ���ʽ����ƽ



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�������и����е������Ʊ����ռ�������Ӧ�����壬�ܲ�����ͼװ�õ���
��Ũ��ˮ����NaOH��NH3 �ڴ���ʯ��ϡ������CO2
�۹���������Һ�Ͷ���������O2��ϡ�����ͭƬ��NO
��Ũ����Ͷ���������Cl2 ��ʯ��ˮ��C2H2
|
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ŨCaCl2��Һ��ͨ��NH3��CO2�������Ƶ�����̼��ƣ�����ֱ����1~10nm֮�䣩����ͼ��ʾA~EΪʵ���ҳ���������װ�ã����̶ֹ��г�װ����ȥ���������Ҫ��ش����⣮
��1��ʵ�����Ʊ�NH3�ķ�Ӧ����ʽ��_______________________________��
��2��ʵ������ȡ���ռ������NH3����ѡ����������װ�õĽӿ�����˳���ǣ�ѡ����ĸ����a�� �� �� �� ��h��
��3����ŨCaCl2��Һ��ͨ��NH3��CO2����������̼���ʱ��Ӧ��ͨ��������� ����д��������̼��Ƶ����ӷ���ʽ ��
��4����ŨCaCl2��Һ��NH3������ȷ������£�CO2�����������ᵼ������̼��Ʋ����½�����CO2������Һ�д������ڵ������У�������������ʵĵ��������ˮ��������������ӣ�________________,
��5��ȡ��Ӧ��ȥ����CaCO3����Һ�ֱ�������ʵ�飬����ʵ���жϺ������ǣ�_________��
| A���μ�����Na2CO3��Һ�����г���˵��CO2һ������ |
| B���μ��������ᣬ�������ݣ�CO2һ������ |
| C��������ҺpH��������7��CO2һ�������� |
| D���μ�����BaCl2��Һ����������CO2һ��û�й��� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��ҵ�Ͽ���ʳ�κ�ʯ��ʯΪ��Ҫԭ�ϣ�����ͬ�ķ������������ش��������⣺
��1��¬����������ʳ�Ρ�ʯ��ʯ��Ũ���ᡢ��̿Ϊԭ�ϣ��ڸ����½������գ��ٽ�ȡ���ᾧ���Ƶô��
��ʳ�κ�Ũ���ᷴӦ�Ļ�ѧ����ʽΪ___________��
�������ƺͽ�̿��ʯ��ʯ��Ӧ�Ļ�ѧ����ʽΪ_________����֪����֮һΪCaS����
��2������Ĺ�������ͼ��ʾ���õ���̼�����ƾ��������ɴ��
| | |
| |  |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��ʴ���
��15�֣�����ʯ���ʵĸ���Ʒ����������(Na2SiF6)���Ʊ���ʯ(Na3AlF6)������ʯ�ǵ���������ۼ�,�ɽ������������۵㡣��ͼ�ǹ�ҵ����������ͼ��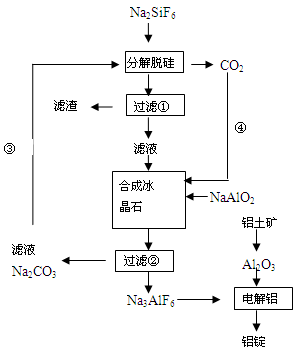
��1����ҵ�ϴ��������Ʊ��ϸߴ���Al2O3����Ҫ����������__________�����ڣ���һ����Ӧ�ķ���ʽ______________________________________________________________
��2�����Ʊ������������ι��˲��������˲����ٵ���Һ��________��Һ��������________ ��
��3���ֽ��ѹ�ͺϳɱ���ʯ��ѧ��Ӧ����ʽ�ֱ�Ϊ��_________________��____________________��
��4�����չ����Тۺܵ͢�Ŀ����_____________________��̼���ƺͶ�����̼�Ƿ��� ��
��5�����Al2O3��Alʱ��I=200kA��һ����Al 1��430 t�����Ч���Ƕ��٣�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
������������ɫ����ˮ����ζ��Һ�壬�е�Ϊ77.2 �棬ʵ����ij����ȡ���ñ�����14.3 mL��95%�Ҵ� 23 mL�����õ�Ũ���ᡢ����̼�����Լ��������Ҵ���ϳ���ˮ������Ȼ�����Һ����Ҫװ����ͼ��ʾ�� 
ʵ�鲽�裺
������A�е�������ƿ��ע�������Ҵ���Ũ�����ҡ�ȣ��ٽ�ʣ�µ������Ҵ��ͱ�����ע���Һ©������á���ʱ��Һ©�����������Ҵ������ʵ���֮��ԼΪ5��7��
�ڼ�����ԡ����Լ135 �桫145��
�۽���Һ©���е�Һ�建������������ƿ����ڼ�������ʹ��������������������ʴ�����ȣ�ֱ��������ɡ�
�ܱ�����ԡ�¶�һ��ʱ�䣬��������Һ�������ֹͣ���ȡ�
��ȡ��B�е���ƿ����һ��������Na2CO3��Һ����������εؼӵ����Һ��ӱ����������ݲ���Ϊֹ��
���ݵ�Һ�������Һ����ȥˮ�㡣
�߽�����CaCl2��Һ�����������뵽��Һ©���У���һ��ʱ����ã��ų�ˮ�㣨��Һ����
���Һ©����õ����dz����ᴿ������������Ʒ��
�Իش�
��1��ʵ����Ũ�������Ҫ������__________ _______��
��2���ù����Ҵ�����ҪĿ����________ _________��
��3���ñ���Na2CO3��Һϴ�Ӵ�����Ŀ����________ ___��
�����NaOH��Һ����Na2CO3��Һ������ĺ��___________________________________��
��4���ñ���CaCl2��Һϴ�Ӵ�����Ŀ����_____ ______��
��5���ڲ�������õĴ����ﻹ���е�������______ ___��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
(12��)SnSO4��һ����Ҫ�������Σ��ڹ�ҵ���������Ź㷺��Ӧ�á����Ʊ�·�����£�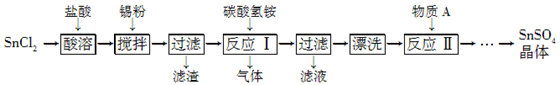
��֪�������������£���Һ�е�Sn2���ɱ������е�����������Sn4���� SnCl2��ˮ�����ɼ�ʽ�Ȼ�����[Sn(OH)Cl]��
��1�� д������A�����ƣ�________��
��2�� SnCl2�����������ˮ�ܽ��ԭ����____________________(�û�ѧ����ʽ��ʾ)��
��3�� ���۵������dz�ȥ����ʱ����������Sn4������д������Sn4�������ӷ���ʽ��______________________________��
��4�� ��Ӧ�����ɵij���ΪSnO��д���÷�Ӧ�Ļ�ѧ����ʽ��____���÷�Ӧ���¶���Ҫ������75 �����ҵ�ԭ����____��
��5�� ʵ�����С�Ưϴ��������ʵ�����������____��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
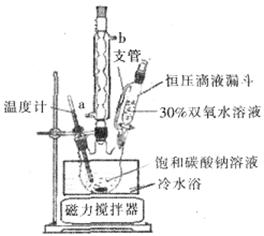
��̼���ƣ�Na2CO3��3H2O2�����й���˫��ˮ���׳ƣ��þ������Na2CO3��H2O2��˫�����ʣ�������Ӧ����ϴ�ӡ�ӡȾ����֯����ֽ��ҽҩ�����������У���̼���Ƶ�ij������������ͼ��ʾ��
��֪��2Na2CO3��3H2O2��2Na2CO3��3H2O ��H��0���ش��������⣺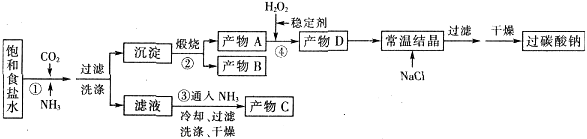
��1���������ʿ�ʹ��̼���ƽϿ�ʧЧ���ǣ�����ţ�_________��
| A��FeCl3��Һ | B��H2S | C��ϡ���� | D��NaHCO3��Һ |
| T/�� | �������ٷֺ��� | ���� |
| 5��10 | 13.94 | 85.49 |
| 10��15 | 14.02 | 85.78 |
| 15��20 | 15.05 | 88.38 |
| 20��25 | 14.46 | 83.01 |
 ��
���鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ʽ̼��ͭ��һ�ֻ���ԭ�ϣ���ѧʽ��mCu(OH)2��nCuCO3��ʾ��ʵ�����Է�ͭмΪԭ����ȡ��ʽ̼��ͭ�IJ������£�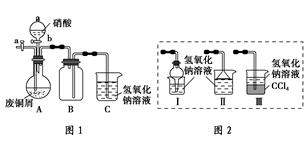
��.��ͭм������ͭ
����1����ͭм�ڿ����г�����գ�������������ϡ���
����2����ͼ1(�г�������ʡ��)����Ũ���Ỻ���ӵ���ͭм��(��ͭм����)����ַ�Ӧ����ˣ��õ�����ͭ��Һ��
����3��������2��Ũ���ỻ��ϡ���ᣬ�������䡣
��.��ʽ̼��ͭ���Ʊ�
������Թ��м���̼������Һ������ͭ��Һ
��ˮԡ������70 ������
����0.4 mol��L��1��NaOH��Һ����pH��8.5�������á�����
������ˮϴ�ӡ���ɣ��õ���ʽ̼��ͭ��Ʒ
��ش��������⣺
(1)������1ʵ�飬����ѡ�������������________(�����)��
(2)ͼ2���ֱܷ����ͼ1��B��Cװ�õ���________(��װ�����)��
(3)��֪��NO��NO2��2NaOH===2NaNO2��H2O��2NO2��2NaOH===NaNO3��NaNO2��H2O��NO���ܵ�����NaOH��Һ��Ӧ��ʵ�����ʱ����β�������ʹװ���е��ж����屻NaOH��Һ��ȫ���գ�__________________________��
(4)�������ϴ�ӵ�Ŀ����______________________________________��
(5)����۹��˺����Һ�к���CO32��������CO32���ķ�����_________________________________________________________��
(6)�ⶨ��ʽ̼��ͭ��ɵķ�����Ҫ�����֣�
����1�����շ���ȡ34.6 g������mCu(OH)2��nCuCO3����Ӳ���Թ������գ��������������ͨ��������Ũ���ᡢ�����ļ�ʯ���У���ȫ���պ�Ũ���Ά��1.8 g����ʯ�Ҿ���8.8 g��
����2����ԭ�����������м�ǿ�ȣ��ⷴӦǰ������������
�������������������ʽ̼��ͭ�Ļ�ѧʽ_____________________��
����ƽ��ѧ����ʽ��mCu(OH)2��nCuCO3��________H2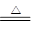 ________Cu��________CO2����________H2O
________Cu��________CO2����________H2O
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com