
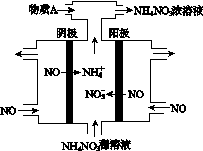
NH3��һϵ�з�Ӧ���Եõ�HNO3��NH4NO3������ͼ��ʾ��
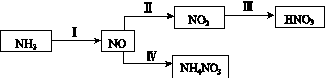
(1)���У�NH3��O2�ڴ��������·�Ӧ���仯ѧ����ʽ��_________________________��
(2)����2NO(g)��O2(g)  2NO2(g)��������������
2NO2(g)��������������
ͬʱ���ֱ���NO��ƽ��ת�����ڲ�ͬѹǿ(p1��p2)�����¶ȱ仯������(��ͼ)��

�ٱȽ�p1��p2�Ĵ�С��ϵ��________��
�����¶����ߣ��÷�Ӧƽ�ⳣ���仯��������________��
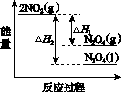
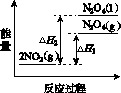
(3)���У������¶ȣ���NO2(g)ת��ΪN2O4(l)�����Ʊ�Ũ���ᡣ
����֪��2NO2(g)  N2O4(g)����H1 2NO2(g)
N2O4(g)����H1 2NO2(g) N2O4(l)����H2
N2O4(l)����H2
���������仯ʾ��ͼ�У���ȷ����(ѡ����ĸ)________��
 ��
��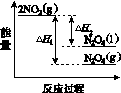
������A������������������B C
��N2O4��O2��H2O���ϵĻ�ѧ����ʽ��________________________________________��
(4)���У����NO�Ʊ�NH4NO3���乤��ԭ����ͼ��ʾ��Ϊʹ������ȫ��ת��ΪNH4NO3���貹��A��A��________��˵�����ɣ�________________________________________��
(1)4NH3��5O2 4NO��6H2O
4NO��6H2O
(2)��p1��p2���ڼ�С
(3)��A����2N2O4��O2��2H2O===4HNO3
(4)NH3�����ݷ�Ӧ��8NO��7H2O 3NH4NO3��2HNO3����������HNO3��
3NH4NO3��2HNO3����������HNO3��
[����] (1)���Ĵ������ķ�Ӧ����ʽΪ
4NH3��5O2 4NO��6H2O��
4NO��6H2O��
(2)����2NO(g)��O2(g)  2NO2(g)��֪�÷�ӦΪ���������С�ķ�Ӧ���¶���ͬ������ѹǿ��ƽ�������ƶ���NO��ƽ��ת����������ͼʾ�����꺬�壬�ж�p1<p2�����ٿ�ͬһѹǿ�ߣ��¶����ߣ�NO��ƽ��ת���ʽ��ͣ�ƽ�����淴Ӧ�����ƶ���������ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�ⳣ����С��
2NO2(g)��֪�÷�ӦΪ���������С�ķ�Ӧ���¶���ͬ������ѹǿ��ƽ�������ƶ���NO��ƽ��ת����������ͼʾ�����꺬�壬�ж�p1<p2�����ٿ�ͬһѹǿ�ߣ��¶����ߣ�NO��ƽ��ת���ʽ��ͣ�ƽ�����淴Ӧ�����ƶ���������ӦΪ���ȷ�Ӧ���¶����ߣ�ƽ�ⳣ����С��
(3)�ٽ����¶ȣ�NO2(g)ת��ΪN2O4(l)����H2<0����Ӧ����������������������C���� N2O4(g)ת��ΪN2O4(l)��Ҫ�ų�����������NO2(g)ת��ΪN2O4(g)��NO2(g)ת��ΪN2O4(l)�ų��������٣�B����������ȷ��ͼʾΪA����N2O4��O2��H2O��Ӧ��������ķ�Ӧ����ʽΪ2N2O4�� O2��2H2O===4HNO3��
(4)���ݹ���ԭ��װ��ͼ������ȷ������ΪNOʧȥ����ת��ΪNO ������NOת��ΪNH
������NOת��ΪNH �����ݵ缫��Ӧ��д�缫��ӦʽΪ��
�����ݵ缫��Ӧ��д�缫��ӦʽΪ��
������NO��3e����2H2O===NO ��4H��
��4H��
������NO��5e���� 6H��===NH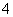 �� H2O
�� H2O
Ȼ����ݵ�ʧ�����غ㣬������������ʵ�����笠��������ʵ����࣬������Ҫ����Һ�м��������ΪNH3(��8NO��7H2O 3NH4NO3��2HNO3)��
3NH4NO3��2HNO3)��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
һ���¶��£�10 mL 0.40 mol/L H2O2��Һ�������ֽ⡣��ͬʱ�̲������O2�����(������Ϊ��״��)���±���
| t/min | 0 | 2 | 4 | 6 | 8 | 10 |
| V(O2)/mL | 0.0 | 9.9 | 17.2 | 22.4 | 26.5 | 29.9 |
������������ȷ����(��Һ����仯���Բ���)(����)
A��0��6 min��ƽ����Ӧ���ʣ�
v(H2O2)��3.3��10��2mol��L��1��min��1
B��0��6 min��ƽ����Ӧ���ʣ�
v(H2O2)<3.3��10��2mol��L��1��min��1
C����Ӧ��6 minʱ��c(H2O2)��0.30 mol/L
D����Ӧ��6 minʱ��H2O2�ֽ���50%
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������Ȼ�����Ƶ���H2��CO��Ϊ��Ҫ��ɵĹ�ҵԭ�Ϻϳ�������ӦΪCH4(g)��H2O(g)
 CO(g)��3H2(g)��
CO(g)��3H2(g)��
(1)������ˮ������Ӧ����������Ԫ����____________�������ɱ�״����35.84 L�ϳ���ʱת�Ƶ��ӵ����ʵ�����________��
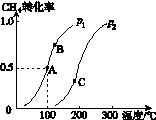
(2)��2 mol CH4��5 mol H2O(g)ͨ���ݻ�Ϊ100 L�ķ�Ӧ�ң�CH4��ƽ��ת�������¶ȡ�ѹǿ�Ĺ�ϵ��ͼK235��ʾ��
ͼK235
�����ﵽA�������ʱ��Ϊ5 min����v(H2)��________________________________________________________________________��
100 ��ʱƽ�ⳣ��K��____________________��
��ͼ�е�p1______p2(�<����>������)��A��B��C�����ƽ�ⳣ��KA��KB��KC�Ĵ�С��ϵ��________________________________________________________________________��
(3)�ϳ������ںϳɰ���ʱ���ȥCO��������ӦCO(g)��H2O(g) 
 CO2(g)��H2(g)����H<0�����д�ʩ����ʹ
CO2(g)��H2(g)����H<0�����д�ʩ����ʹ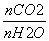 �������________(ѡ����)��
�������________(ѡ����)��
A�������¶�
B�����º����³���He(g)
C����H2����ϵ�з���
D����ͨ��һ������ˮ����
����̼�����Һ�������ɵ�CO2��������pH��10��̼�����Һ����ˮ�����OH�������ʵ���Ũ��Ϊ________________________________________________________________________��
�����£�0.1 mol��L��1 KHCO3��Һ��pH>8������Һ��c(H2CO3)________c(CO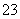 )(�>����������<��)��
)(�>����������<��)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
��֪��
C(s)��O2(g)===CO2(g)����H1
CO2(g)��C(s)===2CO(g)����H2
2CO(g)��O2(g)===2CO2(g)����H3
4Fe(s)��3O2(g)===2Fe2O3(s)����H4
3CO(g)��Fe2O3(s)===3CO2(g)��2Fe(s)����H5
���й���������Ӧ�ʱ���ж���ȷ����(����)
A����H1>0����H3<0 B����H2>0����H4>0
C����H1����H2����H3 D����H3����H4����H5
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
MgAgCl�����һ���ܱ���ˮ�����һ����������أ���ط�Ӧ����ʽΪ2AgCl�� Mg === Mg2���� 2Ag ��2Cl�����йظõ�ص�˵����ȷ����(����)
A��MgΪ��ص�����
B��������ӦΪAgCl��e��===Ag��Cl��
C�����ܱ�KCl ��Һ����
D�������ں���Ӧ����������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������(NiMH)Ŀǰ�Ѿ���Ϊ��϶���������һ����Ҫ������ͣ�NiMH�е�M��ʾ���������Ͻ𣬸õ���ڳ������е��ܷ�Ӧ����ʽ��Ni(OH)2��M===NiOOH��MH��
��֪��6NiOOH��NH3��H2O��OH��===6Ni(OH)2��NO
����˵����ȷ����(����)
A��NiMH��طŵ�����У������ĵ缫��ӦʽΪNiOOH��H2O��e��===Ni(OH)2��OH��
B����������OH��������������Ǩ��
C���������������ĵ缫��Ӧʽ��H2O��M��e��===MH��OH����H2O�е�H��M��ԭ
D��NiMH����п�����KOH��Һ����ˮ����Ϊ�������Һ
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����ˮ������Ũ��ˮ��Դ������������ۺ����ú�ˮ����Ҫ;��֮һ��һ�����Ƚ���ˮ������õ�ˮ���ٴ�ʣ���Ũ��ˮ��ͨ��һϵ�й���������ȡ������Ʒ��
�ش��������⣺
(1)���иĽ����Ż���ˮ�ۺ����ù��յ�������������е���________(�����)��
���û�������ȡ��ˮ
����߲��ֲ�Ʒ������
���Ż���ȡ��Ʒ��Ʒ��
�ܸĽ��ء��塢þ�ȵ���ȡ����
(2)���á���������������Ũ��ˮ����Br2�����ô������ա������������Ҫ��Ӧ��Br2��Na2CO3��H2O��NaBr��NaBrO3��NaHCO3������1 mol Br2ʱ��ת�Ƶĵ�����Ϊ________mol��
(3)��ˮ��þ��һ�ι�����������ͼ��

Ũ��ˮ����Ҫ�ɷ����£�
| ���� | Na�� | Mg2�� | Cl�� | SO |
| Ũ��/(g��L��1) | 63.7 | 28.8 | 144.6 | 46.4 |
�ù��չ����У��������Ҫ��Ӧ�����ӷ���ʽΪ______________________________����Ʒ2�Ļ�ѧʽΪ__________��1 LŨ��ˮ���ɵõ���Ʒ2������Ϊ________g��
(4)����ʯī���������������������ڵ��Ȼ�þ��������Ӧ�Ļ�ѧ����ʽΪ________________________�����ʱ����������ˮ���ڻ���ɲ�Ʒþ�����ģ�д���йط�Ӧ�Ļ�ѧ����ʽ��____________________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
������ʵ����ȷ����(����)
A������װ�г����������������Ϊ��ԭ�����ۡ��Ȼ��ơ�̿�۵ȣ�������ԭ���������������ʴ��ͬ����������������ԭ��ص��������缫��ӦΪFe��2e��===Fe2��
B����������ȼ�ϵ�ص������缫��ӦΪ2H2O��O2��4e��===4OH��
C����ʯī�缫���CuCl2��Һ�������Ϸ����ķ�ӦΪ2Cl����2e��===Cl2��
D��Ǧ���ص�������ӦΪPbO2��4H����2e��===Pb2����2H2O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
���������������(����)
A��SO2ʹ��ˮ��ɫ����ϩʹKMnO4��Һ��ɫ��ԭ����ͬ
B���Ʊ���������ʱ�����ȵ�NaOH��Һ�ռ������Գ�ȥ���е�����
C���ñ���ʳ��ˮ���ˮ����ʯ��Ӧ�����Լ�����Ȳ�IJ�������
D����AgNO3��Һ���Լ���KCl��KI
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com