
 Al2O3+2Fe
Al2O3+2Fe
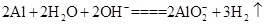
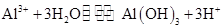
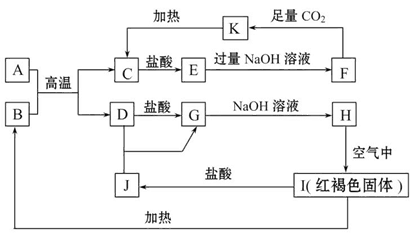
 C+D��֪��AΪAl��CΪAl2O3����EΪAlCl3��FΪNaAlO2��KΪAl��OH��3��
C+D��֪��AΪAl��CΪAl2O3����EΪAlCl3��FΪNaAlO2��KΪAl��OH��3��

 ��Ȥ����¹�֪��ϵ�д�
��Ȥ����¹�֪��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��MgO��Al2O3��SiO2�ֱ��Ǽ����������������������������� |
| B�����߾����Բ��õ����ڿ�����ȼ�ջ���ȵķ����Ƶ� |
| C��MgO��Al2O3������Ϊ���²��ϣ�SiO2�������������ά |
| D���ֱ�������״̬��MgO��Al2O3��SiO2�����Ƶ�Mg��Al��Si |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
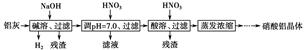
| Al��OH��3 | ��ʼ���� | ������ȫ | ������ʼ�ܽ� | �����ܽ���ȫ |
| pH | 3.3 | 5.0 | 7.8 | 12.8 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����������ˮ |
| B�������������������� |
| C���������������Ũ���� |
| D����������������Ƶ����� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
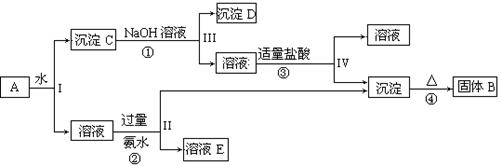
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��Al2O3��NaOH | B��Al��Fe3O4 | C��Al��CuSO4 | D��Al��HCl |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��3��4 | B��9��32 | C��9��16 | D��9��8 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com