
ГЦШЁ2.379 gН№ЪєЛЏКЯЮяЃЈЛЏбЇЪНЮЊMaXb?cH2OЃЉЃЌгУЙ§СПSOCl2ЃЈbp
ЭбЫЎЗДгІЪНЃКSOCl2 + H2O = SO2 + 2 HCl
Лђ MaXb?cH2O + cSOCl2 = MaXb + c SO2 + 2c HCl
SO2БЛH2O2бѕЛЏГЩSO42ЃЃЌГСЕэМСЮЊBaCl2ЃЌЕУ14.004 g BaSO4ЃЈSжЪСПЗжЪ§ЮЊ13.74%ЃЉ
MЃЈЃЉ= 32.07/13.74 = 233.4 ЛђгУЯрЖддзгСПМЦЫувВПЩ
14.004 g/233.0ЃЈg/molЃЉ= 0.060 mol МДбљЦЗжаКЌга0.060 mol ЕФH2O
0.060 mol ЁС 18.0 g/mol = 1.080 gЃЈH2OЃЉ
бљЦЗжаMaXbжЪСПЮЊ 2.379 g 1.080 g = 1.299 g
гЩДЫЧѓЕУЃК1.190 g MaXb?cH2OжаКЌMaXb жЪСПЮЊ1.299ЁС1.019/2.379 = 0.650 g
20 mLЪдвКжаКЌMaXbЮЊ0.650 g ¸ 5 = 0.130 g
МгAgNO3ЕФАзЩЋГСЕэЮЊAgClЃЌЦфЮяжЪЕФСПЮЊ0.287/143.4 = 2 ЁС 10Ѓ3 mol
ЫЕУїMaXbжаКЌCl ЮЊ0.071 g
MaXb жаMжЪСПЮЊ0.130Ѓ0.071 = 0.059 gЃЌClЮЊ0.071 gЃЌгЩдзгСПБэПЩжЊMaXb ЮЊCoCl2
дЛЏКЯЮяЮЊCoCl2?6H2O



| ФъМЖ | ИпжаПЮГЬ | ФъМЖ | ГѕжаПЮГЬ |
| ИпвЛ | ИпвЛУтЗбПЮГЬЭЦМіЃЁ | ГѕвЛ | ГѕвЛУтЗбПЮГЬЭЦМіЃЁ |
| ИпЖў | ИпЖўУтЗбПЮГЬЭЦМіЃЁ | ГѕЖў | ГѕЖўУтЗбПЮГЬЭЦМіЃЁ |
| ИпШ§ | ИпШ§УтЗбПЮГЬЭЦМіЃЁ | ГѕШ§ | ГѕШ§УтЗбПЮГЬЭЦМіЃЁ |
ПЦФПЃКИпжаЛЏбЇ РДдДЃК2012НьЩЯКЃЪаЫЩНЧјИпШ§ЩЯбЇЦкЦкФЉЃЈ1дТЃЉПМЪдЛЏбЇЪдОэЃЈДјНтЮіЃЉ ЬтаЭЃКМЦЫуЬт
ЃЈБОЬтЙВ16ЗжЃЉЬњбѕЬхЪЧвЛРрДХадВФСЯЃЌЭЈЪНЮЊMOЁЄxFe2O3ЃЈMЮЊЖўМлН№ЪєРызгЃЉЃЌШчFe3O4ПЩБэЪОЮЊFeOЁЄFe2O3ЃЌжЦБИСїГЬЮЊЃК
ИљОнЬтвтЛиД№ЃК
1ЃЎЯжГЦШЁ2.8 gЬњЗлЃЌМгШывЛЖЈСПЕФЯЁСђЫсЪЙЦфЭъШЋШмНтКѓЃЌдйМгШы gЕФFeCl3ЙЬЬхЃЌОЩЯЪіЗДгІКѓЃЌЗНПЩЕУЕНДПОЛЕФFe3O4ЁЃ
2ЃЎШчЙћвдMgSO4КЭFeCl3ЮЊдСЯЃЌвЊЛёЕУ18.0ПЫMgOЁЄ2Fe2O3ЃЌдкжЦШЁЙ§ГЬжажСЩйашвЊ1 mol/LЕФNaOHШмвК mLЁЃ
3ЃЎдквЛДЮжЦБИFe3O4ЕФЪЕбщжаЃЌгЩгкЗДгІЮяБШР§ПижЦВЛЕБЃЌЛёЕУСЫСэвЛжжВњЦЗЁЃШЁвЛЖЈСПИУВњЦЗШмгкзуСПбЮЫсжаЃЌЛЙашЭЈШыБъзМзДПіЯТ672 mLCl2ВХФмАбШмвКжаЕФFe2+ШЋВПбѕЛЏЮЊFe3+ЃЌШЛКѓАбЫљЕУШмвКеєИЩЃЌзЦЩежСКужиЃЌЕУ9.6 gЙЬЬхЁЃИУВњЦЗЕФЛЏбЇЪНЮЊ ЁЃ
4ЃЎгавЛжждкЕчзгЙЄвЕЩЯгІгУЙуЗКЕФКЌИѕЬњбѕЬхЃЌЦфзщГЩПЩБэЪОЮЊЃК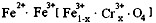 ЃЌЯжга15.2 g FeSO4ЃЌОCr2O72-ЁЂH2O2ЕШЮяжЪДІРэКѓЃЈВЛдйв§ШыКЌЬњЮяжЪЃЉЃЌПЩЕУЕНКЌИѕЬњбѕЬхЕФжЪСПЗЖЮЇЮЊ ЁЃ
ЃЌЯжга15.2 g FeSO4ЃЌОCr2O72-ЁЂH2O2ЕШЮяжЪДІРэКѓЃЈВЛдйв§ШыКЌЬњЮяжЪЃЉЃЌПЩЕУЕНКЌИѕЬњбѕЬхЕФжЪСПЗЖЮЇЮЊ ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК2013-2014бЇФъИпПМЛЏбЇЖўТжзЈЬтИДЯА ГЃМћН№ЪєдЊЫиЕЅжЪМАЦфживЊЛЏКЯЮяСЗЯАОэЃЈНтЮіАцЃЉ ЬтаЭЃКЬюПеЬт
ЬњЁЂТСЁЂЭЕШН№ЪєМАЦфЛЏКЯЮядкШеГЃЩњЛюжагІгУЙуЗК,ЧыИљОнЯТСаЪЕбщЛиД№ЮЪЬт:
(1)ЩњЬњжаКЌгавЛжжЬњЬМЛЏКЯЮяX(Fe3C)ЁЃXдкзуСПЕФПеЦјжаИпЮТьбЩе,ЩњГЩгаДХадЕФЙЬЬхY,НЋYШмгкЙ§СПбЮЫсЕФШмвКжаКЌгаЕФДѓСПбєРызгЪЧЁЁЁЁЁЁЁЃ
(2)ФГШмвКжагаMg2+ЁЂFe2+ЁЂAl3+ЁЂCu2+ЕШЫФжжРызг,ЯђЦфжаМгШыЙ§СПЕФNaOHШмвККѓ,Й§ТЫ,НЋТЫдќИпЮТзЦЩеВЂНЋзЦЩеКѓЕФЙЬЬхЭЖШыЙ§СПЕФЯЁбЮЫсжа,ЫљЕУШмвКгыдШмвКЯрБШ,ШмвКжаДѓСПМѕЩйЕФбєРызгЪЧЁЁЁЁЁЁЁЃ
A.Mg2+B.Fe2+??????????????????? C.Al3+D.Cu2+
(3)бѕЛЏЬњЪЧживЊЙЄвЕбеСЯ,гУЗЯЬњаМжЦБИЫќЕФСїГЬШчЯТ:
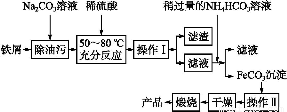
ЛиД№ЯТСаЮЪЬт:
ЂйВйзїЂёЕФУћГЦЪЧЁЁЁЁЁЁ,ВйзїЂђЕФУћГЦЪЧЁЁЁЁЁЁЁЃ
ЂкаДГідкПеЦјжаьбЩеFeCO3ЕФЛЏбЇЗНГЬЪНЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЃ
(4)гааЉЭЌбЇШЯЮЊKMnO4ШмвКЕЮЖЈвВФмНјааЬњдЊЫиКЌСПЕФВтЖЈЁЃ
a.ГЦШЁ2.850 gТЬЗЏ(FeSO4ЁЄ7H2O)ВњЦЗ,ШмНт,дк250 mLШнСПЦПжаЖЈШн;
b.СПШЁ25.00 mLД§ВтШмвКЗХШызЖаЮЦПжа;
c.гУСђЫсЫсЛЏЕФ0.010 00 molЁЄL-1 KMnO4ШмвКЕЮЖЈжСжеЕу,ЯћКФKMnO4ШмвКЬхЛ§ЕФЦНОљжЕЮЊ20.00 mLЁЃ
ЂйЪЕбщЧА,ЪзЯШвЊОЋШЗХфжЦвЛЖЈЮяжЪЕФСПХЈЖШЕФKMnO4ШмвК250 mL,ХфжЦЪБашвЊЕФвЧЦїГ§ЬьЦНЁЂВЃСЇАєЁЂЩеБЁЂНКЭЗЕЮЙмЭт,ЛЙашЁЁЁЁЁЁЁЃ
ЂкФГЭЌбЇЩшМЦЕФЯТСаЕЮЖЈЗНЪН,зюКЯРэЕФЪЧЁЁЁЁЁЁЁЃ(МаГжВПЗжТдШЅ)(ЬюзжФИађКХ)
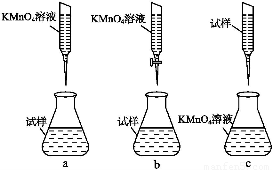
ЂлаДГіЕЮЖЈЙ§ГЬжаЗДгІЕФРызгЗНГЬЪН:ЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЃ
ЂмМЦЫуЩЯЪібљЦЗжаFeSO4ЁЄ7H2OЕФжЪСПЗжЪ§ЮЊЁЁЁЁЁЁЁЁЁЁЁЁЁЁЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК2013-2014бЇФъеуНЪЁКМжнЪаИпШ§ЕкЖўДЮдТПМЛЏбЇЪдОэЃЈНтЮіАцЃЉ ЬтаЭЃКЪЕбщЬт
ФГбЇЩњЖдSO2гыЦЏЗлОЋЕФЗДгІНјааЪЕбщЬНОПЃК
Вйзї | ЯжЯѓ |
ШЁЪЪСПЦЏЗлОЋЙЬЬхЃЌМгШы100mLЫЎ | ВПЗжЙЬЬхШмНтЃЌШмвКТдгабеЩЋ |
Й§ТЫЃЌВтЦЏЗлОЋШмвКЕФpH | pHЪджНЯШБфРЖЃЌКѓЭЪЩЋ |
| вКУцЩЯЗНГіЯжАзЮэЃЛ ЩдКѓЃЌГіЯжЛызЧЃЌШмвКБфЮЊЛЦТЬЩЋЃЛ ЩдКѓЃЌВњЩњДѓСПАзЩЋГСЕэЃЌЛЦТЬЩЋЭЪШЅ |
ЃЈ1ЃЉаДГіC12КЭCa(OH)2жЦШЁЦЏЗлОЋЕФЛЏбЇЗНГЬЪН?????????????????????????????? ЃЛ???
ЃЈ2ЃЉЯђЫЎжаГжајЭЈШыSO2ЃЌЮДЙлВьЕНАзЮэЁЃЭЦВтЯжЯѓiЕФАзЮэгЩHClаЁвКЕЮаЮГЩЃЌНјааШчЯТЪЕбщЃК
a.гУЪЊШѓЕФЕтЛЏМиЕэЗлЪджНМьбщАзЮэЃЌЮоБфЛЏЃЛ
b.гУЫсЛЏЕФAgNO3ШмвКМьбщАзЮэЃЌВњЩњАзЩЋГСЕэЁЃ
ЂйЪЕбщaФПЕФЪЧ???????????????????????????????????????? ЃЛ
ЂкгЩЪЕбщaЁЂbВЛФмХаЖЯАзЮэжаКЌгаHC1ЃЌРэгЩЪЧ?????????????????????????????????????? ЃЛ
ЃЈ3ЃЉНЋAЦПжаЛьКЯЮяЙ§ТЫЁЂЯДЕгЃЌЕУЕНГСЕэX
ЂйЯђГСЕэXжаМгШыЯЁHC1ЃЌЮоУїЯдБфЛЏЁЃШЁЩЯВуЧхвКЃЌМгШыBaC12ШмвКЃЌВњЩњАзЩЋГСЕэЁЃдђГСЕэXжаКЌгаЕФЮяжЪЪЧ???????? ЃЛ
ЂкгУРызгЗНГЬЪННтЪЭЯжЯѓiiiжаЛЦТЬЩЋЭЪШЅЕФдвђЃК??????????????????????????????????? ЁЃ
ЃЈ4ЃЉВтЖЈЦЏЗлОЋгааЇГЩЗжЕФжЪСПЗжЪ§ЁЃГЦШЁ2.000 gЦЏЗлОЋгкзЖаЮЦПжаЃЌМгЫЎШмНтЃЌЕїНкШмвКЕФpHЃЌвдЕэЗлЮЊжИЪОМСЃЌгУ0.2000 molЁЄL-1 KIШмвКНјааЕЮЖЈЃЌШмвКГіЯжЮШЖЈЧГРЖЩЋЪБЮЊЕЮЖЈжеЕуЁЃЗДгІдРэЮЊЃК3C1O-+ I- == 3C1Ѓ+ IO3-
IO3- + 5IЃ + 3H2O == 6OH-+ 3I2
ЪЕбщВтЕУЪ§ОнШчЯТБэЫљЪОЁЃ
ЕЮЖЈДЮЪ§ | 1 | 2 | 3 |
KIШмвКЬхЛ§/mL | 19.98 | 20.02 | 20.00 |
ИУЦЏАзЗлжагааЇГЩЗжЕФжЪСПЗжЪ§ЮЊ????????????????????????? ЁЃ
ВщПДД№АИКЭНтЮі>>
ПЦФПЃКИпжаЛЏбЇ РДдДЃК2011-2012бЇФъЩЯКЃЪаЫЩНЧјИпШ§ЩЯбЇЦкЦкФЉЃЈ1дТЃЉПМЪдЛЏбЇЪдОэЃЈНтЮіАцЃЉ ЬтаЭЃКМЦЫуЬт
ЃЈБОЬтЙВ16ЗжЃЉЬњбѕЬхЪЧвЛРрДХадВФСЯЃЌЭЈЪНЮЊMOЁЄxFe2O3ЃЈMЮЊЖўМлН№ЪєРызгЃЉЃЌШчFe3O4ПЩБэЪОЮЊFeOЁЄFe2O3ЃЌжЦБИСїГЬЮЊЃК
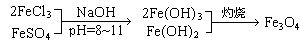
ИљОнЬтвтЛиД№ЃК
1ЃЎЯжГЦШЁ2.8 gЬњЗлЃЌМгШывЛЖЈСПЕФЯЁСђЫсЪЙЦфЭъШЋШмНтКѓЃЌдйМгШы gЕФFeCl3ЙЬЬхЃЌОЩЯЪіЗДгІКѓЃЌЗНПЩЕУЕНДПОЛЕФFe3O4ЁЃ
2ЃЎШчЙћвдMgSO4КЭFeCl3ЮЊдСЯЃЌвЊЛёЕУ18.0ПЫMgOЁЄ2Fe2O3ЃЌдкжЦШЁЙ§ГЬжажСЩйашвЊ1 mol/LЕФNaOHШмвК mLЁЃ
3ЃЎдквЛДЮжЦБИFe3O4ЕФЪЕбщжаЃЌгЩгкЗДгІЮяБШР§ПижЦВЛЕБЃЌЛёЕУСЫСэвЛжжВњЦЗЁЃШЁвЛЖЈСПИУВњЦЗШмгкзуСПбЮЫсжаЃЌЛЙашЭЈШыБъзМзДПіЯТ672 mLCl2ВХФмАбШмвКжаЕФFe2+ШЋВПбѕЛЏЮЊFe3+ЃЌШЛКѓАбЫљЕУШмвКеєИЩЃЌзЦЩежСКужиЃЌЕУ9.6 gЙЬЬхЁЃИУВњЦЗЕФЛЏбЇЪНЮЊ ЁЃ
4ЃЎгавЛжждкЕчзгЙЄвЕЩЯгІгУЙуЗКЕФКЌИѕЬњбѕЬхЃЌЦфзщГЩПЩБэЪОЮЊЃК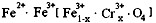 ЃЌЯжга15.2
g
FeSO4ЃЌОCr2O72-ЁЂH2O2ЕШЮяжЪДІРэКѓЃЈВЛдйв§ШыКЌЬњЮяжЪЃЉЃЌПЩЕУЕНКЌИѕЬњбѕЬхЕФжЪСПЗЖЮЇЮЊ
ЁЃ
ЃЌЯжга15.2
g
FeSO4ЃЌОCr2O72-ЁЂH2O2ЕШЮяжЪДІРэКѓЃЈВЛдйв§ШыКЌЬњЮяжЪЃЉЃЌПЩЕУЕНКЌИѕЬњбѕЬхЕФжЪСПЗЖЮЇЮЊ
ЁЃ
ВщПДД№АИКЭНтЮі>>
АйЖШжТаХ - СЗЯАВсСаБэ - ЪдЬтСаБэ
КўББЪЁЛЅСЊЭјЮЅЗЈКЭВЛСМаХЯЂОйБЈЦНЬЈ | ЭјЩЯгаКІаХЯЂОйБЈзЈЧј | ЕчаХеЉЦОйБЈзЈЧј | ЩцРњЪЗащЮожївхгаКІаХЯЂОйБЈзЈЧј | ЩцЦѓЧжШЈОйБЈзЈЧј
ЮЅЗЈКЭВЛСМаХЯЂОйБЈЕчЛАЃК027-86699610 ОйБЈгЪЯфЃК58377363@163.com