
ĆŗµÄĘų»ÆŹĒøߊ§”¢Ēå½ąµŲĄūÓĆĆŗĢæµÄÖŲŅŖĶ¾¾¶Ö®Ņ»”£
(1)ŌŚ250C 101kPaŹ±£¬H2ÓėO2»ÆŗĻÉś³É1mol H2O(g)·Å³ö241.8kJµÄČČĮ棬ĘäČČ»Æѧ·½³ĢŹ½ĪŖ
___________
ÓÖÖŖ: ¢ŁC(s)£«O2(g)ØTCO2(g) ”÷H£½£393.5kJ/mol
¢ŚCO(g)£«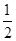 O2(g)ØTCO2(g) ”÷H£½£283.0kJ/mol
O2(g)ØTCO2(g) ”÷H£½£283.0kJ/mol
½¹ĢæÓėĖ®ÕōĘų·“Ó¦ŹĒ½«¹ĢĢåĆŗ±äĪŖĘųĢåČ¼ĮĻµÄ·½·Ø£¬C(s)£«H2O(g)ØTCO(g)£«H2(g) ”÷H=____kJ/mol
(2) COæÉŅŌÓėH2O(g)½ųŅ»²½·¢Éś·“Ó¦: CO(g)£«H2O(g)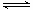 CO2(g)£«H2(g) ”÷H£¼0ŌŚŗćČŻĆܱÕČŻĘ÷ÖŠ£¬ĘšŹ¼Ź±n(H2O)=0.20mol£¬n(CO)£½0.10 mol,ŌŚ8000CŹ±“ļµ½Ę½ŗāדĢ¬£¬K£½1.0£¬ŌņĘ½ŗāŹ±£¬ČŻĘ÷ÖŠCOµÄ×Ŗ»ÆĀŹŹĒ_____________(¼ĘĖć½į¹ū±£ĮōŅ»Ī»Š”Źż)”£
CO2(g)£«H2(g) ”÷H£¼0ŌŚŗćČŻĆܱÕČŻĘ÷ÖŠ£¬ĘšŹ¼Ź±n(H2O)=0.20mol£¬n(CO)£½0.10 mol,ŌŚ8000CŹ±“ļµ½Ę½ŗāדĢ¬£¬K£½1.0£¬ŌņĘ½ŗāŹ±£¬ČŻĘ÷ÖŠCOµÄ×Ŗ»ÆĀŹŹĒ_____________(¼ĘĖć½į¹ū±£ĮōŅ»Ī»Š”Źż)”£
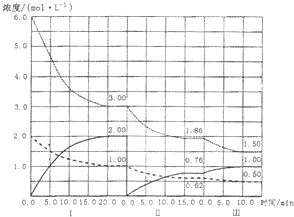
(3) ¹¤ŅµÉĻ“ÓĆŗĘų»ÆŗóµÄ»ģŗĻĪļÖŠ·ÖĄė³öH2£¬½ųŠŠ°±µÄŗĻ³É£¬ŅŃÖŖ·“Ó¦·“Ó¦N2(g)£«3H2(g 2NH3(g)£Ø”÷H£¼0£©ŌŚµČČŻĢõ¼žĻĀ½ųŠŠ£¬øıäĘäĖū·“Ó¦Ģõ¼ž£¬ŌŚI”¢II”¢III½×¶ĪĢåĻµÖŠø÷ĪļÖŹÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĻĀĶ¼ĖłŹ¾£ŗ
2NH3(g)£Ø”÷H£¼0£©ŌŚµČČŻĢõ¼žĻĀ½ųŠŠ£¬øıäĘäĖū·“Ó¦Ģõ¼ž£¬ŌŚI”¢II”¢III½×¶ĪĢåĻµÖŠø÷ĪļÖŹÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĻĀĶ¼ĖłŹ¾£ŗ

¢ŁN2µÄĘ½¾ł·“Ó¦ĖŁĀŹv1(N2)”¢vII(N2)”¢vIII(N2)“ӓ󵽊”ÅÅĮŠ“ĪŠņĪŖ________£»
¢ŚÓɵŚŅ»“ĪĘ½ŗāµ½µŚ¶ž“ĪĘ½ŗā£¬Ę½ŗāŅĘ¶ÆµÄ·½Ļņ ŹĒ________£¬²ÉČ”µÄ“ėŹ©ŹĒ________”£
¢Ū±Č½ĻµŚII½×¶Ī·“Ó¦ĪĀ¶Č(T2)ŗĶµŚIII½×¶Ī·“Ó¦ĖŁ¶Č£ØT3)µÄøßµĶ£ŗT2________T3Ģī”°”µ”¢=”¢<”±ÅŠ¶ĻµÄĄķÓÉŹĒ________________”£
£Ø14·Ö£©£Ø1£©H2(g)+ 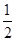 O2(g)ØTlH2O(g) ”÷H£½£241.8kJ/mol £Ø2·Ö£©£»£«131.3kJ £Ø1·Ö£©
O2(g)ØTlH2O(g) ”÷H£½£241.8kJ/mol £Ø2·Ö£©£»£«131.3kJ £Ø1·Ö£©
£Ø2£©66.7% £Ø2·Ö£© £Ø3£©¢Łv1(N2)£¾vII(N2)£¾vIII(N2) £Ø3·Ö£©
¢ŚĻņÕż·“Ó¦·½Ļņ “Ó·“Ó¦ĢåĻµÖŠŅĘ³ö²śĪļNH3£Ø3·Ö£©
¢Ū£¾ “Ė·“Ó¦ĪŖ·ÅČČ·“Ó¦£¬½µµĶĪĀ¶Č£¬Ę½ŗāĻņÕż·“Ó¦·½ĻņŅĘ¶Æ £Ø3·Ö£©
”¾½āĪö”æ
ŹŌĢā·ÖĪö£ŗ£Ø1£©ŌŚ25”ę”¢101kPaŹ±£¬H2ÓėO2»ÆŗĻÉś³É1molH2O(g)·Å³ö241.8kJµÄČČĮ棬ĖłŅŌĘäČČ»Æѧ·½³ĢŹ½ĪŖH2(g)+ 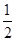 O2(g)ØTlH2O(g) ”÷H£½£241.8kJ/mol”£
O2(g)ØTlH2O(g) ”÷H£½£241.8kJ/mol”£
ŅŃÖŖ¢ŁC(s)£«O2(g)ØTCO2(g) ”÷H£½£393.5kJ/mol
¢ŚCO(g)£«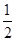 O2(g)ØTCO2(g) ”÷H£½£283.0kJ/mol
O2(g)ØTCO2(g) ”÷H£½£283.0kJ/mol
¢ŪH2(g)+ 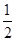 O2(g)ØTlH2O(g) ”÷H£½£241.8kJ/mol
O2(g)ØTlH2O(g) ”÷H£½£241.8kJ/mol
ĖłŅŌøł¾ŻøĒĖ¹¶ØĀÉ£¬¢Ł£¢Ū£¢Ś¼“µĆµ½C(s)£«H2O(g)ØTCO(g)£«H2(g)£¬Ōņ·“Ó¦ČČ”÷H£½£393.5kJ/mol£«241.8kJ/mol£«283.0kJ/mol£½£«131.3kJ/mol”£
£Ø2£©
CO(g)£«H2O(g) CO2(g)£«H2(g)
CO2(g)£«H2(g)
ĘšŹ¼Įæ£Ømol£© 0.1 0.2 0 0
×Ŗ»ÆĮæ£Ømol£© x x x x
Ę½ŗāĮæ£Ømol£© 0.1£x 0.2£x x x
ÓÉÓŚ·“Ó¦Ē°ŗóĘųĢåµÄĢå»ż²»±ä£¬æÉŅŌÓĆĪļÖŹµÄĮæ“śĢęÅØ¶Č¼ĘĖćĘ½ŗā³£Źż
¼“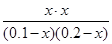 £½1.0
£½1.0
½āµĆx£½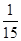
ĖłŅŌĘ½ŗāŹ±£¬ČŻĘ÷ÖŠCOµÄ×Ŗ»ÆĀŹŹĒ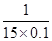 ”Į100%£½66.7%
”Į100%£½66.7%
£Ø3£©¢Łøł¾ŻĶ¼ĻńæÉÖŖ£¬ŠéĻß±ķŹ¾µŖĘųµÄÅØ¶Č±ä»Æ£¬Ōņv1(N2)£½£Ø2mol/L£1mol/L£©”Ā20min£½0.05mol/£ØL•min£©£¬vII(N2)£½£Ø1mol/L£0.62mol/L£©”Ā15min£½0.0253mol/£ØL•min£©£¬vIII(N2)£½£Ø0.62mol/L£0.5mol/L£©”Ā10min£½0.012mol/£ØL•min£©£¬¹ŹN2µÄĘ½¾ł·“Ó¦ĖŁĀŹv1(N2)£¾vII(N2)£¾vIII(N2).
¢Śøł¾ŻĶ¼ĻńæÉÖŖ£¬µŚ¢ņ½×¶Ī°±ĘųŹĒ“Ó0æŖŹ¼µÄ£¬Ė²¼ä·“Ó¦ĪļµŖĘųŗĶĒāĘųÅØ¶Č²»±ä£¬Ņņ“ĖæÉŅŌČ·¶ØµŚŅ»“ĪĘ½ŗāŗó“ÓĢåĻµÖŠŅĘ³öĮĖ°±Ęų£¬¼“¼õÉŁÉś³ÉĪļÅØ¶Č£¬Ę½ŗāÕżĻņŅĘ¶Æ”£
¢ŪµŚ¢ó½×¶ĪµÄæŖŹ¼ÓėµŚ¢ņ½×¶ĪµÄĘ½ŗāø÷ĪļÖŹµÄĮæ¾łĻąµČ£¬øł¾Ż°±ĘųŗĶĒāĘųµÄĮæ¼õÉŁ£¬°±ĘųµÄĮæŌö¼ÓæÉÅŠ¶ĻĘ½ŗāŹĒÕżĻņŅĘ¶ÆµÄ”£øł¾ŻĘ½ŗāæŖŹ¼Ź±ÅضČČ·¶Ø“ĖĘ½ŗāŅĘ¶Æ²»æÉÄÜŹĒÓÉÅØ¶ČµÄ±ä»ÆŅżĘšµÄ£¬ĮķĶāĢāÄæĖłøųĢõ¼žČŻĘ÷µÄĢå»ż²»±ä£¬ŌņøıäŃ¹ĒæŅ²²»æÉÄÜ£¬Ņņ“ĖŅ»¶ØĪŖĪĀ¶ČµÄÓ°Ļģ”£“Ė·“Ó¦ÕżĻņĪŖ·ÅČČ·“Ó¦£¬æÉŅŌĶĘ²āĪŖ½µµĶĪĀ¶Č£¬Ņņ“Ė“ļµ½Ę½ŗāŗóĪĀ¶ČŅ»¶Ø±ČµŚ¢ņ½×¶ĪĘ½ŗāŹ±µÄĪĀ¶ČµĶ£¬¼“T2£¾T3”£
æ¼µć£ŗæ¼²éČČ»Æѧ·½³ĢŹ½µÄŹéŠ“£»øĒĖ¹¶ØĀɵÄÓ¦ÓĆ£»·“Ó¦ĖŁĀŹŗĶĘ½ŗā³£ŹżµÄ¼ĘĖćŅŌ¼°Ķā½ēĢõ¼ž¶Ō·“Ó¦ĖŁĀŹŗĶĘ½ŗāדĢ¬µÄÓ°ĻģµČ



| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| 1 |
| 2 |
| 1 |
| 2 |
| 1 |
| 2 |
| ĘšŹ¼ĪļÖŹµÄĮæ | ¼× | ŅŅ | ±ū |
| n£ØH2O£©/mol | 0.10 | 0.20 | 0.20 |
| n£ØCO£©/mol | 0.10 | 0.10 | 0.20 |
| c(CO2)”Įc(H2) |
| c(CO)”Įc(H2O) |
| c(CO2)”Įc(H2) |
| c(CO)”Įc(H2O) |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
 £Ø2012?ŃĢĢØŅ»Ä££©ĆŗµÄĘų»ÆŹĒøߊ§”¢Ēå½ąµÄĄūÓĆĆŗĢæµÄÖŲŅŖĶ¾¾¶Ö®Ņ»£®
£Ø2012?ŃĢĢØŅ»Ä££©ĆŗµÄĘų»ÆŹĒøߊ§”¢Ēå½ąµÄĄūÓĆĆŗĢæµÄÖŲŅŖĶ¾¾¶Ö®Ņ»£®| 1 |
| 2 |
| 1 |
| 2 |
| H2O | CO | CO2 | H2 | |
| ¼× n/mol | 0.10 | 0.30 | 0.10 | 0.50 |
| ŅŅ n/mol | 0.20 | 0.20 | 0 | 0 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗ
| 1 |
| 2 |
| “߻ƼĮ |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗŌĶĮĄķ½ā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ2013-2014ѧğŗžÄĻŹ””°ĪåŹŠŹ®Š£”±øßČż12ŌĀĮŖŗĻ¼ģ²ā»ÆѧŹŌ¾ķ£Ø½āĪö°ę£© ĢāŠĶ£ŗĢīæÕĢā
¹ż¶ČÅÅ·ÅCO2»įŌģ³É”°ĪĀŹŅŠ§Ó¦”±£¬ĪŖĮĖ¼õÉŁĆŗČ¼ÉÕ¶Ō»·¾³Ōģ³ÉµÄĪŪČ¾£¬ĆŗµÄĘų»ÆŹĒøߊ§”¢Ēå½ąĄūÓĆĆŗĢæµÄÖŲŅŖĶ¾¾¶”£Ćŗ×ŪŗĻĄūÓƵÄŅ»ÖÖĶ¾¾¶ČēĶ¼ĖłŹ¾”£

£Ø1£©ŅŃÖŖ¢ŁC(s) £« H2O(g) = CO(g)£«H2(g) ¦¤H1£½£«131.3 kJ”¤mol£1
¢ŚC(s) £« 2H2O(g) = CO2(g) £« 2H2(g) ¦¤H2£½£«90 kJ”¤mol£1
ŌņŅ»Ńõ»ÆĢ¼ÓėĖ®ÕōĘų·“Ӧɜ³É¶žŃõ»ÆĢ¼ŗĶĒāĘųµÄČČ»Æѧ·½³ĢŹ½ŹĒ ________________________£¬
£Ø2£©ÓĆĻĀĶ¼Ōµē³Ų×°ÖĆæÉŅŌĶź³É¹ż³Ģ¢ŻµÄ×Ŗ»Æ£¬øĆ×°ÖĆbµē¼«µÄµē¼«·“Ó¦Ź½ŹĒ_______________________”£

£Ø3£©ŌŚŃ¹ĒæĪŖ0.1 MPaĢõ¼žĻĀ£¬ČŻ»żĪŖV LµÄĆܱÕČŻĘ÷ÖŠa mol COÓė2a mol H2ŌŚ“߻ƼĮ×÷ÓĆĻĀ·“Ӧɜ³É¼×“¼£ŗ
CO(g)£«2H2(g)  CH3OH(g)£¬COµÄĘ½ŗā×Ŗ»ÆĀŹÓėĪĀ¶Č”¢Ń¹ĒæµÄ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£¬Ōņ£ŗ
CH3OH(g)£¬COµÄĘ½ŗā×Ŗ»ÆĀŹÓėĪĀ¶Č”¢Ń¹ĒæµÄ¹ŲĻµČēĻĀĶ¼ĖłŹ¾£¬Ōņ£ŗ

¢Łp1________p2(Ģī”°£¾”±”¢”°£¼”±»ņ”°£½”±)”£
¢ŚŌŚĘäĖūĢõ¼ž²»±äµÄĒéæöĻĀ£¬ĻņČŻĘ÷ÖŠŌŁŌö¼Óa mol COÓė2a mol H2£¬“ļµ½ŠĀĘ½ŗāŹ±£¬COµÄĘ½ŗā×Ŗ»ÆĀŹ________(Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£
¢ŪŌŚp1ĻĀ£¬100 ”ꏱ£¬CO(g)£«2H2(g)
 CH3OH(g)·“Ó¦µÄĘ½ŗā³£ŹżĪŖ________(ÓĆŗ¬a”¢VµÄ“śŹżŹ½±ķŹ¾)”£
CH3OH(g)·“Ó¦µÄĘ½ŗā³£ŹżĪŖ________(ÓĆŗ¬a”¢VµÄ“śŹżŹ½±ķŹ¾)”£
£Ø4£©ČēĶ¼±ķŹ¾CO2ÓėH2·“Ӧɜ³ÉCH3OHŗĶH2OµÄ¹ż³ĢÖŠÄÜĮæ(µ„Ī»ĪŖkJ”¤mol£1)µÄ±ä»Æ£ŗ

¹ŲÓŚøĆ·“Ó¦µÄĻĀĮŠĖµ·ØÖŠ£¬ÕżČ·µÄŹĒ________(Ģī±ąŗÅ)”£
A£®¦¤H£¾0£¬¦¤S£¾0 B£®¦¤H£¾0£¬¦¤S£¼0
C£®¦¤H£¼0£¬¦¤S£¼0 D£®¦¤H£¼0£¬¦¤S£¾0
£Ø5£©ĪŖĢ½¾æ·“Ó¦ŌĄķ£¬ĻÖ½ųŠŠČēĻĀŹµŃ飬ŌŚĢå»żĪŖ1 LµÄĆܱÕČŻĘ÷ÖŠ£¬³äČė1 mol
CO2ŗĶ3 mol H2£¬Ņ»¶ØĢõ¼žĻĀ·¢Éś·“Ó¦£ŗCO2(g)£«3H2(g)
 CH3OH(g)£«H2O(g)£¬²āµĆCO2(g)ŗĶCH3OH(g)µÄÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĶ¼ĖłŹ¾£ŗ
CH3OH(g)£«H2O(g)£¬²āµĆCO2(g)ŗĶCH3OH(g)µÄÅضČĖꏱ¼ä±ä»ÆµÄĒśĻßČēĶ¼ĖłŹ¾£ŗ

¢Ł“Ó·“Ó¦æŖŹ¼µ½Ę½ŗā£¬CO2µÄĘ½¾ł·“Ó¦ĖŁĀŹv(CO2)£½________”£
¢ŚĻĀĮŠ“ėŹ©ÖŠÄÜŹ¹»ÆŃ§Ę½ŗāĻņÕż·“Ó¦·½ĻņŅĘ¶ÆµÄŹĒ________(Ģī±ąŗÅ)”£
A£®ÉżøßĪĀ¶Č B£®½«CH3OH(g)¼°Ź±Ņŗ»ÆŅĘ³ö
C£®Ń”Ōńøߊ§“߻ƼĮ D£®ŌŁ³äČė1 mol CO2ŗĶ3 mol H2
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com