
�ڻ�ҩ���й��Ŵ��Ĵ���֮һ�����ı�ը��ӦΪ��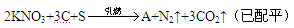
(1)�������г��˵����������̼�⣬����һ������A���ɣ���A�ĵ���ʽΪ ��A���� ���塣
(2)�ڻ�ҩ��λ��Ԫ�����ڱ������ڵ�Ԫ���� �֡�����һ��Ԫ�صĵ��ʿ�������ҩ���������������Ư��֯��ͷ���������Ԫ�������ڱ��е�λ���� ����Ԫ�ص�ԭ�Ӻ����� �ֲ�ͬ�˶�״̬�ĵ��ӡ�
(3)�ڻ�ҩ������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳���� (��Ԫ�ط��ű�ʾ)��
(4)������ʵ��˵���ڻ�ҩ��̼������Ԫ�طǽ��������ǿ������ ��
| A��ͬ����ͬŨ����ҺpH��Na2CO3��Na2SO4 | B�����ԣ�H2SO3��H2CO3 |
| C��CS2��̼Ԫ��Ϊ+4�ۣ���Ԫ��Ϊ-2�� | D���ֽ��¶ȣ�CH4��H2S |
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�ڻ�ҩ���й��Ŵ��Ĵ���֮һ�����ı�ը��ӦΪ��
(1)�������г��˵����������̼�⣬����һ������A���ɣ���A�ĵ���ʽΪ ��A���� ���塣
(2)�ڻ�ҩ��λ��Ԫ�����ڱ������ڵ�Ԫ���� �֡�����һ��Ԫ�صĵ��ʿ�������ҩ���������������Ư��֯��ͷ���������Ԫ�������ڱ��е�λ���� ����Ԫ�ص�ԭ�Ӻ����� �ֲ�ͬ�˶�״̬�ĵ��ӡ�
(3)�ڻ�ҩ������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳���� (��Ԫ�ط��ű�ʾ)��
(4)������ʵ��˵���ڻ�ҩ��̼������Ԫ�طǽ��������ǿ������ ��
A��ͬ����ͬŨ����ҺpH��Na2CO3��Na2SO4 B�����ԣ�H2SO3��H2CO3
C��CS2��̼Ԫ��Ϊ+4�ۣ���Ԫ��Ϊ-2�� D���ֽ��¶ȣ�CH4��H2S
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013�����ʡ�����и�����ѧ����ĩ���Ի�ѧ�Ծ����������� ���ͣ������
�ڻ�ҩ���й��Ŵ��Ĵ���֮һ�����ı�ը��ӦΪ��
2KNO3+3C+S 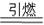 K2S+N2��+3a ����ƽ
K2S+N2��+3a ����ƽ
�ٻ�����ԭ�ӵĽṹʾ��ͼ ��
���������г����������⣬����һ������A���ɣ���A�Ļ�ѧʽΪ ��
�ۺڻ�ҩ��λ��Ԫ�����ڱ������ڵ�Ԫ���� �֣�����һ��Ԫ�ص������������Ư��ֽ����˿����ñ��Ȳ�������ɱ������������Ԫ�������ڱ��е�λ���� ��д����������ͨ��FeCl3��Һ��Ӧ�����ӷ���ʽ ��
��������ʵ��˵���ڻ�ҩ��̼������Ԫ�طǽ��������ǿ������ ��
| A��ͬ����ͬŨ����Һ��pH��H2CO3��H2SO4 | B���ȶ��ԣ�H2SO4��H2CO3 |
| C��CS2��̼Ԫ��Ϊ+4�ۣ���Ԫ��Ϊ-2�� | D���ܽ��ԣ�H2S��CH4 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012-2013ѧ�����ʡ�����и�����ѧ����ĩ���Ի�ѧ�Ծ��������棩 ���ͣ������
�ڻ�ҩ���й��Ŵ��Ĵ���֮һ�����ı�ը��ӦΪ��
2KNO3+3C+S 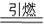 K2S+N2��+3a ����ƽ
K2S+N2��+3a ����ƽ
�ٻ�����ԭ�ӵĽṹʾ��ͼ ��
���������г����������⣬����һ������A���ɣ���A�Ļ�ѧʽΪ ��
�ۺڻ�ҩ��λ��Ԫ�����ڱ������ڵ�Ԫ���� �֣�����һ��Ԫ�ص������������Ư��ֽ����˿����ñ��Ȳ�������ɱ������������Ԫ�������ڱ��е�λ���� ��д����������ͨ��FeCl3��Һ��Ӧ�����ӷ���ʽ ��
��������ʵ��˵���ڻ�ҩ��̼������Ԫ�طǽ��������ǿ������ ��
A��ͬ����ͬŨ����Һ��pH��H2CO3��H2SO4 B���ȶ��ԣ�H2SO4��H2CO3
C��CS2��̼Ԫ��Ϊ+4�ۣ���Ԫ��Ϊ-2�� D���ܽ��ԣ�H2S��CH4
����֪����ʱCuS��Ksp��1.3��10��36����100 ml 2��10��18 mol/L ��K2S��Һ�м���100 ml
2��10��18 mol/L��CuCl2��Һ,��ͨ������˵������CuS�������ɣ�д�������������̣�������Һ���ʱ������仯���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ���Ϻ���������������ѧ����ĩ������⻯ѧ�Ծ� ���ͣ������
�ڻ�ҩ���й��Ŵ��Ĵ���֮һ�����ı�ը��ӦΪ��
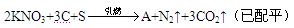
(1)�������г��˵����������̼�⣬����һ������A���ɣ���A�ĵ���ʽΪ ��A���� ���塣
(2)�ڻ�ҩ��λ��Ԫ�����ڱ������ڵ�Ԫ���� �֡�����һ��Ԫ�صĵ��ʿ�������ҩ���������������Ư��֯��ͷ���������Ԫ�������ڱ��е�λ���� ����Ԫ�ص�ԭ�Ӻ����� �ֲ�ͬ�˶�״̬�ĵ��ӡ�
(3)�ڻ�ҩ������Ԫ�ص�ԭ�Ӱ뾶�Ӵ�С��˳���� (��Ԫ�ط��ű�ʾ)��
(4)������ʵ��˵���ڻ�ҩ��̼������Ԫ�طǽ��������ǿ������ ��
A��ͬ����ͬŨ����ҺpH��Na2CO3��Na2SO4 B�����ԣ�H2SO3��H2CO3
C��CS2��̼Ԫ��Ϊ+4�ۣ���Ԫ��Ϊ-2�� D���ֽ��¶ȣ�CH4��H2S
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com