
���������μ����ֽ��Ҳ���ϸ��ӡ�ijѧϰС����Mg(NO3)2Ϊ�о�������ͨ��ʵ��̽�����ȷֽ�IJ���������4�ֲ��룺
�ף�Mg(NO3)2��NO2��O2 �ң�MgO��NO2��O2 ����Mg3N2��O2 ����MgO��NO2��N2
��1��ʵ��ǰ��С���Ա�������϶����붡��������������____ ________��
�������ϵ�֪��2NO2+2NaOH=NaNO3+NaNO2+H2O
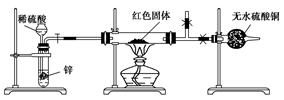
��Լס��ҡ������룬�������ͼ��ʾ��ʵ��װ�ã�ͼ�м��ȡ��г������Ⱦ�ʡ�ԣ���
��2��ʵ�����
��ȡ�����Ӻ��˹����Լ�֮ǰ���ر�k����Ӳ�ʲ����ܣ�A�����۲쵽E �������������ų�������_ _______
�� ��ȡMg(NO3)2����3 . 79 g����A�У�����ǰͨ��N2������װ���ڵĿ�������Ŀ����________���ر�K���þƾ��Ƽ���ʱ����ȷ��������________Ȼ��̶��ڹ��й��岿λ�¼��ȡ�
�� �۲쵽A ���к���ɫ������֣�C��D ��δ�����Ա仯��
�� ����Ʒ��ȫ�ֽ⣬A װ����ȴ�����¡����������ʣ����������Ϊ1.0g
�� ȡ����ʣ��������Թ��У���������ˮ��δ����������
��3��ʵ������������
�� ����ʵ�������ʣ�����������������ɳ���ȷ�ϲ���_______����ȷ�ġ�
�� ����D ������������һλͬѧ��Ϊ����ȷ�Ϸֽ��������O2����Ϊ����O2��D�н�����������ԭ��Ӧ��_________________ ____����д��ѧ����ʽ������Һ��ɫ����ȥ��С�������϶��ֽ��������O2���ڣ�δ��ൽ��ԭ����_____________________��
�� С�����ۺ��ɵĹ�ʶ������ʵ������Բ����ƣ���Ľ�װ���һ���о���
��1��������������ԭ��Ӧԭ����2�֣�
��2����װ�����������ã�2�֣�
�ڱ���Բ���O2�ļ���������ţ�2�֣����ƶ��ƾ���Ԥ��Ӳ�ʲ����ܣ�2�֣�
��3������ ��2�֣� ��2Na2SO3 + O2= 2Na2SO4��2�֣�
O2��ͨ��װ��Bʱ�Ѳ��뷴Ӧ��2�֣�
����������������⿼��Mg(NO)3���ȷֽ��IJ����̽�����Լ�ʵ�����������
��1��Mg(NO3)2��Mg��N��O�Ļ��ϼ۷ֱ�Ϊ��2����5����2�ۣ�NO2��N2��Mg2N3�е��Ļ��ϼ۷ֱ�Ϊ��4��0����3�ۣ�O2�����Ļ��ϼ���0�ۡ��ʼס��ҡ�����Ԫ�صĻ��ϼۡ������н���������������ԭ��Ӧ�ص㡣��������ֻ�л��ϼ۵Ľ��ͣ�û�л��ϼ۵����ߣ��ò²ⲻ������
��2�����һ��װ���������ݣ�˵������װ�����������á�N2��װ���еĿ����ϳ���Ŀ�ģ��Ƿ�ֹ�Էֽ�����O2�ļ��鳬�ɸ��š����Ȳ�������Ԥ�ȣ��ټ��м��ȡ�
��3����Mg(NO3)2������ˮ��Mg3N2�ܹ���ˮ��Ӧ��Mg3N2��6H2O��3Mg(OH)2����2NH3����MgO������ˮ������������ˮ��δ�����������Ҳ�����ȷ��
��Na2SO3Ϊ����ǿ���Σ�SO32-ˮ����Һ�ʼ��ԣ�SO32-��H2O HSO3-��OH-����Һ��ɫ��ȥ��˵�������ܹ�����Na2SO3ΪNa2SO4��2Na2SO3��O2��2Na2SO4��
HSO3-��OH-����Һ��ɫ��ȥ��˵�������ܹ�����Na2SO3ΪNa2SO4��2Na2SO3��O2��2Na2SO4��
δ���鵽��������������NO2��O2��ˮ��ͬ���ÿ��Բ���HNO3����NaOH���գ�����������O2�Ĵ��ڡ�
���㣺���黯ѧʵ������������



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
����ͭ���仯����һ�㶼�����������ɫ����
| Cu | Cu2O | CuO | Cu��OH��2 | CuSO4��5H2O |
| ��ɫ�����Ϻ�ɫ�� | ��ɫ����ש��ɫ�� | ��ɫ | ��ɫ | ��ɫ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�������Ʊ���Ϊ��ҵ�Σ���Ư�ס���Ƶȷ���Ӧ�ù㷺����ľ̿��Ũ���ᡢˮ��ͭΪԭ�����ɵ�һ��������������Ʒ�Ӧ�Ʊ��������Ƶ�װ������ͼ��ʾ�����ּг�װ���ԣ���
��֪�������£���2NO+Na2O2��2NaNO2 ��3NaNO2+3HCl��3NaCl+HNO3+2NO��+H2O��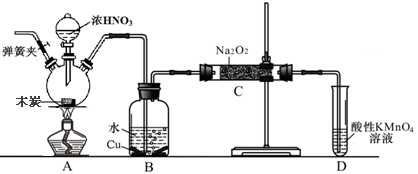
�����������£�NO��NO2�C������MnO4�C��Ӧ����NO3�C��Mn2+
���������գ�
��1��д��Ũ������ľ̿��Ӧ�Ļ�ѧ����ʽ ��
��2��B�й۲쵽����Ҫ������ ��Dװ�õ������� ��
��3������C�в������������Ƶķ����� ��
��4��������C�����г�������������и�����̼���ƺ� ��Ϊ���������Щ������Ӧ��B��Cװ�ü�����װ��E������E��ע��E��ʢ�ŵ�ҩƷ���� ��
��5��д������C�������Ƿ�̼���Ƶķ��� ��
��6����1.56g����������ȫת����Ϊ�������ƣ�������������Ҫľ̿ g��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
ijС�������ͼװ�ã����ּг�װ������ȥ������̽����ʪ��Cl2��Na2CO3��Ӧ�õ��������ʵijɷ֡�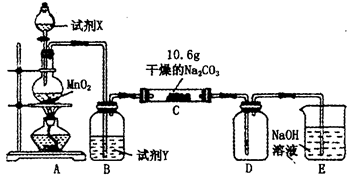
��1��A����ȡCl2�����ӷ���ʽ_______________________________________.
��2��ͨ��һ������ʪ��Cl2��Ӧ����⣬D��ֻ��Cl2Oһ�����壬C��ֻ��һ�������⣬ͬʱ����NaHCO3�ȣ�ijͬѧ��C�����ù�������ijɷֽ���̽����
������������衣
����1���������ֳɷ֣�NaHCO3��_____________________________��
����2���������ֳɷ֣�NaHCO3��_____________________________��
����Ʒ���������ʵ�顣д��ʵ�鲽���Լ�Ԥ������ͽ��ۡ�
��ѡʵ���Լ�������������ˮ��ϡHNO3��BaCl2��Һ������ʯ��ˮ��AgNO3��Һ���Թܡ�С�ձ���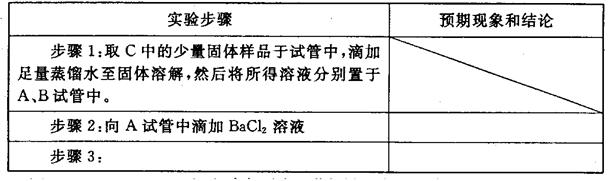
��3����֪C����0��1 mol Cl2�μӷ�Ӧ��������һ����������֪C�з�Ӧ�Ļ�ѧ����ʽΪ__________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
�о���ѧϰС���ij�����������壨FeSO4?xH2O���ȷֽ��о�����С��ͬѧ��ȡag��������������Ʒ��ͼ1���¼��ȣ�ʹ����ȫ�ֽ⣬�����ò������̽������ͨ������װ��B�������x��ֵ��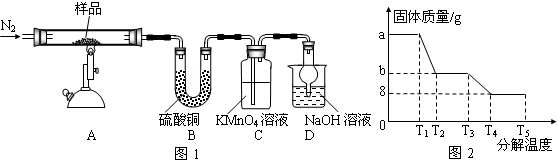
��1��װ��B������ͭ��ĩ��������������12.6g��˵����������ˮ��װ��C�и��������Һ��ɫ��˵�������л��� ��
��2��ʵ����Ҫ����ͨ�뵪������������x�� ���ƫ����ƫС�����䡱����
��3����������������ȫ�ֽ��װ��A�л���������ɫ����Fe2O3��
��4���������Ϸ����ó����������ֽ������һ����SO3��д��FeSO4�ֽ�Ļ�ѧ����ʽ ��
��5��װ��D���θ���ܵ����� ��
��6��ij�о�������SDTQ600�ȷ����Ƕ������������壨FeSO4?xH2O�������ȷֽ⣬���������ݣ����Ƴɹ������������ֽ��¶ȵĹ�ϵͼ��ͼ2������ͼ2���й����ݣ��ɼ����FeSO4?xH2O�е�x= ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
���к�������NaCl��Na2SO4��Na2CO3�����ʵ�NaNO3��Һ��ѡ���ʵ����Լ���ȥ���ʣ��õ�������NaNO3���壬ʵ����������ͼ��ʾ��
��1������A����Ҫ�ɷ��� �� ���ѧʽ����
��2�����з�Ӧ�����ӷ���ʽ�� ��
��3���٢ڢ��о����еķ�������� ��
��4�����м��������Na2CO3��Һ��Ŀ���� ��
��5����Һ3�����������Եõ�NaNO3���壬��Һ3�п϶����е������� ��Ϊ�˳�ȥ���ʣ�������Һ3�м��������� ��
��6��ʵ����������ʵ���õ�NaNO3��������500 mL 0.40 mol/L NaNO3��Һ��
��������Һʱ���������²�����a�����ݣ�b�����㣻c���ܽ⣻d��ҡ�ȣ�e��ת�ƣ�f��ϴ�ӣ�j����������ȡNaNO3����������� g�����ղ���˳��4���� ������ţ���
��ijͬѧת����Һ�IJ�����ͼ��ʾ��ͼ���������������ձ��� ����ͬѧ�����еĴ����� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ij��ѧ�С���������ͼ��ʾ�����ּг�װ������ȥ��ʵ��װ�ã���̽����ʪ��Cl2�����Na2CO3 ���巴Ӧ�õ��Ĺ������ʵijɷ֡�
��֪��ͨ��һ�������������D��ֻ��һ�ֳ�����Ϊ�ƺ�ɫ�����壬��Ϊ�������������ȷ������C�й��庬��NaHCO3 ���Һ��ȵ���ֻ��һ�֡��ֶ�C�ijɷֽ��в����̽����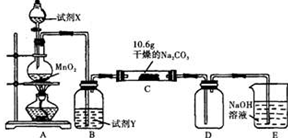
��1������������룺����֪C����0.1molCl2ǡ�ú�10.6��Na2CO3������ȫ��Ӧ����C����Cl2���뷴Ӧ�Ļ�ѧ����ʽ���� ��
��2��������������衣
����1���������ֳɷ֣�NaHCO3�� ��
����2���������ֳɷ֣�NaHCO3�� �� ��
����ƺ���������C�����е�δ֪�ɷֽ���̽������д��ʵ�鲽���Լ�Ԥ������ͽ��ۣ��ɲ���������
��ѡʵ���Լ�������������ˮ��ϡHNO3��Ba(OH)2��Һ��BaCl2��Һ������ʯ��ˮ��AgNO3��Һ���Թܡ�С�ձ���
| ʵ�鲽�� | Ԥ������ͽ��� |
| ����1��ȡ����C�й�����Ʒ���Թ��У�������������ˮ���������������ȫ�ܽ⣬Ȼ��������Һ��װA��B��֧�Թ��С� | |
| ����2�� | |
| ����3�� | |
| | |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
��ˮ�Ǿ����Դ���⣬�Ӻ�ˮ����ȡʳ�κ���Ĺ������£�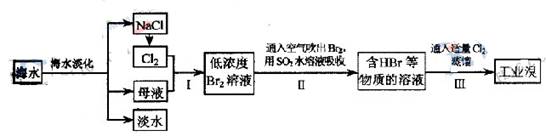
��1����������ѻ��Br2����������ֽ�Br2��ԭΪBr?����Ŀ��Ϊ ��
��2���������SO2ˮ��Һ����Br2�������ʿɴ�95�����йط�Ӧ�����ӷ���ʽΪ ���ɴ˷�Ӧ��֪�������������⣬�ڹ�ҵ������Ӧ�������Ҫ������ ��
��3��ij��ѧ�о���ѧϰС��Ϊ�˽�ӹ�ҵ�����ᴿ��ķ������������й����ϣ�Br2�ķе�Ϊ59�档����ˮ���ж��Ժ�ǿ��ʴ�ԡ����Dzι��������̺�������װ�ü�ͼ��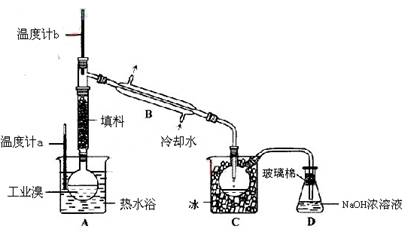
�������������ۣ�
��ͼ������B������ ��
������ʵ��װ�����������Ӿ��������������ܣ���ԭ���� ��
��ʵ��װ�����������ã�Ҫ�ﵽ�ᴿ���Ŀ�ģ���������ο��ƹؼ������� ��
��C��Һ�������ɫΪ ��Ϊ��ȥ�ò������Բ���������Cl2���������м���NaBr��Һ����ַ�Ӧ���ٽ��еķ�������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ʵ����
I��ij��ѧ����С�����Ⱦ�����IJ��ַǽ������������̽�����������ĿҪ��ش��������⡣
(1)д����������ȡNO�����ӷ���ʽ ��
(2)�����ϵ�֪��HCOOH CO+H2O��ʵ��������ͼl��ʾ��װ�ã���ȡCO�����ѡ�õ�װ��Ϊ
CO+H2O��ʵ��������ͼl��ʾ��װ�ã���ȡCO�����ѡ�õ�װ��Ϊ
(�����)��ʵ�������ø�װ�û�����ȡ�ij��������� ��дһ������ķ���ʽ����
(3)�����ϵ�֪�����ô�����ʹ����β���е�һ����̼�͵�������ַ�����Ӧת��Ϊ������̼�͵�������С����ʵ����ģ������β���������������ͼ2��ʾװ��(���ּгֺ�װ������ȥ)��

��ʵ��ǰ���ر�����K����ͨ�����ž�װ���еĿ�������Ŀ���� ��
��װ��(III)����Ҫ������ ��
�۸���װ�����в�����֮������Ӧ��װ��(��)�� װ�á�
II���ÿ���С����ƵĴ�ZnSO4��FeCl3�Ļ����Һ����ȡZnSO4��7H2O�������£�
a���ڻ��Һ�м���6 mol/L NaOH��Һ����pH=8Ϊֹ��
b�����˺�õ�������������ˮ���ϴ�ӳ�����
c����ϴ�Ӻ�ij����м���2 mol/L�����ᣬ������Һ��pH��4��6��������У����ȹ��ˣ���Һ��ΪZnSO4��Һ��
d����Һ�м���2 mol/L�����ᣬʹ��pH=2��
��֪�����������������������ʽ��ʼ��������ȫ����ʱ��Һ��pH���±����ش��������⣺
| ������ | Fe(OH)3 | Zn(OH)2 |
| pH | 1.5��3.2 | 6.4��8.0 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com