
£Ø14·Ö£©ĒāÄÜŹĒÖŲŅŖµÄŠĀÄÜŌ“”£“¢Ēā×÷ĪŖĒāÄÜĄūÓĆµÄ¹Ų¼ü¼¼Źõ£¬ŹĒµ±Ē°¹Ų×¢µÄČȵćÖ®Ņ»”£
£Ø1£©ĒāĘųŹĒĒå½ąÄÜŌ“£¬ĘäČ¼ÉÕ²śĪļĪŖ__________”£
£Ø2£©NaBH4ŹĒŅ»ÖÖÖŲŅŖµÄ“¢ĒāŌŲĢ壬ÄÜÓėĖ®·“Ó¦“ļµ½NaBO2£¬ĒŅ·“Ó¦Ē°ŗóBµÄ»ÆŗĻ¼Ū²»±ä£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ___________£¬·“Ó¦ĻūŗÄ1mol NaBH4Ź±×ŖŅʵĵē×ÓŹżÄæĪŖ__________”£
£Ø3£©“¢Ēā»¹æɽčÖśÓŠ»śĪļ£¬ČēĄūÓĆ»·¼ŗĶéŗĶ±½Ö®¼äµÄæÉÄę·“Ó¦Ą“ŹµĻÖĶŃĒāŗĶ¼ÓĒā£ŗ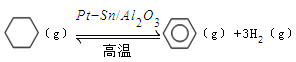 ”£Ä³ĪĀ¶ČĻĀ£¬ĻņŗćČŻĆܱÕČŻĘ÷ÖŠ¼ÓČė»·¼ŗĶé£¬ĘšŹ¼ÅضČĪŖa mol/L£¬Ę½ŗāŹ±±½µÄÅضČĪŖbmol/L£¬øĆ·“Ó¦µÄĘ½ŗā³£ŹżK£½_____”£
”£Ä³ĪĀ¶ČĻĀ£¬ĻņŗćČŻĆܱÕČŻĘ÷ÖŠ¼ÓČė»·¼ŗĶé£¬ĘšŹ¼ÅضČĪŖa mol/L£¬Ę½ŗāŹ±±½µÄÅضČĪŖbmol/L£¬øĆ·“Ó¦µÄĘ½ŗā³£ŹżK£½_____”£
£Ø4£©Ņ»¶ØĢõ¼žĻĀ£¬Ģā11Ķ¼Ź¾×°ÖĆæÉŹµĻÖÓŠ»śĪļµÄµē»Æѧ“¢Ēā£ØŗöĀŌĘäĖüÓŠ»śĪļ£©”£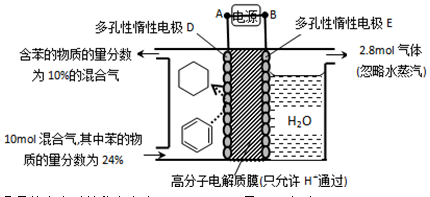
¢Łµ¼ĻßÖŠµē×ÓŅĘ¶Æ·½ĻņĪŖ____________”£
¢ŚÉś³ÉÄæ±ź²śĪļµÄµē¼«·“Ó¦Ź½ĪŖ_________”£
¢ŪøĆ“¢Ēā×°ÖƵĵēĮ÷Š§ĀŹ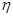 £½_____£Ø
£½_____£Ø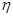 £½
£½ ”Į100%£¬¼ĘĖć½į¹ū±£ĮōŠ”Źżµćŗó1Ī»£©
”Į100%£¬¼ĘĖć½į¹ū±£ĮōŠ”Źżµćŗó1Ī»£©
£Ø1£©Ė®»ņH2O £Ø2£©NaBH4£«2H2O=NaBO2£«4H2”ü£»4NA»ņ2.408”Į1024
£Ø3£©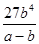 mol3/L3 £Ø4£©¢ŁA”śD ¢ŚC6H6£«6H£«£«6e££½C6H12 ¢Ū64.3%
mol3/L3 £Ø4£©¢ŁA”śD ¢ŚC6H6£«6H£«£«6e££½C6H12 ¢Ū64.3%
½āĪöŹŌĢā·ÖĪö£ŗ£Ø1£©ĒāĘųµÄČ¼ÉÕ²śĪļŹĒĖ®”£
£Ø2£©·“Ó¦Ē°ŗóBŌŖĖŲµÄ»ÆŗĻ¼Ū²»±ä£¬Ōņ·“Ó¦Ē°ŗóBŌŖĖŲµÄ»ÆŗĻ¼Ū¾łŹĒ£«3¼Ū£¬Ņņ“Ė·“Ó¦Ē°NaBH4ÖŠĒāŌŖĖŲµÄ»ÆŗĻ¼ŪŹĒ£1¼Ū”£Ė®ÖŠĒāŌŖĖŲµÄ»ÆŗĻ¼ŪŹĒ£«1¼Ū£¬Ņņ“Ė·“Ó¦ÖŠ»¹ÓŠĒāĘųÉś³É£¬Ōņ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖNaBH4£«2H2O=NaBO2£«4H2”ü”£NaBH4ÖŠĒāŌŖĖŲµÄ»ÆŗĻ¼Ū“Ó£1¼ŪÉżøßµ½0¼Ū£¬Ņņ“Ė1molNaBH4ŌŚ·“Ó¦ÖŠŹ§Č„4molµē×Ó£¬Ę䏿ÄæŹĒ4NA»ņ2.408”Į1024”£
£Ø3£©Ę½ŗāŹ±±½µÄÅØ¶ČŹĒb mol/L£¬Ōņøł¾Ż·“Ó¦µÄ·½³ĢŹ½æÉÖŖĻūŗÄ»·ĪģĶéµÄÅØ¶ČŹĒb mol/L£¬Éś³ÉĒāĘųµÄÅØ¶ČŹĒ3 b mol/L£¬£¬Ę½ŗāŹ±»·ĪģĶéµÄÅضČĪŖ£Øa£b£©mol/L”£ÓÉÓŚ»ÆŃ§Ę½ŗā³£ŹżŹĒŌŚŅ»¶ØĢõ¼žĻĀ£¬µ±æÉÄę·“Ó¦“ļµ½Ę½ŗāדĢ¬Ź±£¬Éś³ÉĪļÅØ¶ČµÄĆŻÖ®»żŗĶ·“Ó¦ĪļÅØ¶ČµÄĆŻÖ®»żµÄ±ČÖµ£¬ŌņøĆĪĀ¶ČĻĀ·“Ó¦µÄĘ½ŗā³£ŹżĪŖ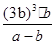 £½
£½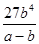 mol3/L3”£
mol3/L3ӣ
£Ø4£©¢Ł±½Éś³É»·ĪģĶéŹōÓŚµĆĒā·“Ó¦£¬Ņņ“ĖŹĒ»¹Ō·“Ó¦£¬¼“µē¼«DŹĒŅõ¼«£¬µē¼«EŹĒŃō¼«£¬Ņņ“Ėµ¼ĻßÖŠµē×ÓµÄĮ÷¶Æ·½ĻņŹĒA”śD”£
¢Ś±½µĆµ½µē×ÓÉś³É»·ĪģĶéŹĒÄæ±ź²śĪļ£¬ÓÉÓŚ“ęŌŚÖŹ×Ó½»»»Ä¤£¬ĖłŅŌĒāĄė×ÓĻņŅõ¼«ŅĘ¶Æ£¬Ōņµē¼«·“Ó¦Ź½ĪŖC6H6£«6H£«£«6e££½C6H12”£
¢ŪŃō¼«Éś³É2.8molĘųĢ壬øĆĘųĢåÓ¦øĆŹĒŃō¼«OH£·ÅµēÉś³ÉµÄŃõĘų£¬Ōņ×ŖŅʵē×ÓµÄĪļÖŹµÄĮ棽2.8mol”Į4£½11.2mol”£ÉčŅõ¼«Ļūŗı½µÄĪļÖŹµÄĮæŹĒxmol£¬ŌņĶ¬Ź±Éś³É xmol»·ĪģĶ飬øł¾Żµē¼«·“Ó¦Ź½C6H6£«6H£«£«6e££½C6H12æÉÖŖµĆµ½µē×ÓŹĒ6xmol£¬øł¾Żµē×ÓŹŲŗćæÉÖŖ£¬Ņõ¼«Éś³ÉĒāĘųŹĒ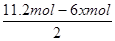 £½5.6mol£3xmol£¬ĖłŅŌ
£½5.6mol£3xmol£¬ĖłŅŌ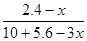 £½0.1£¬½āµĆx£½1.2£¬Ņņ“Ė“¢Ēā×°ÖƵĵēĮ÷Š§ĀŹ£½
£½0.1£¬½āµĆx£½1.2£¬Ņņ“Ė“¢Ēā×°ÖƵĵēĮ÷Š§ĀŹ£½ ”Į100%£½64.3%”£
”Į100%£½64.3%”£
æ¼µć£ŗæ¼²éŃõ»Æ»¹Ō·“Ó¦·½³ĢŹ½ÅäĘ½ŗĶ¼ĘĖć”¢Ę½ŗā³£Źż¼ĘĖćŅŌ¼°µē»ÆѧŌĄķµÄÓ¦ÓĆÓė¼ĘĖć


 °ŁÄźŃ§µäæĪŹ±Ń§Į·²āĻµĮŠ“š°ø
°ŁÄźŃ§µäæĪŹ±Ń§Į·²āĻµĮŠ“š°ø ČŹ°®Ó¢ÓļĶ¬²½Į·Ļ°²įĻµĮŠ“š°ø
ČŹ°®Ó¢ÓļĶ¬²½Į·Ļ°²įĻµĮŠ“š°ø
| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø8·Ö£©£Ø1£©ĒāŹĒĪ“Ą“×īŗƵÄÄÜŌ“Ń”Ōń£¬ÖĘČ”ĒāĘųµÄ³ÉŹģµÄ·½·ØÓŠŗܶą£¬ĄūÓĆ¼×“¼æÉŅŌÓėĖ®ÕōĘų·“Ӧɜ³ÉĒāĘų£¬·“Ó¦·½³ĢŹ½ČēĻĀ£ŗ
CH3OH(g) + H2O(g)  CO2(g) + 3H2(g)
CO2(g) + 3H2(g) 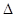 H(298K)£½+ 49.4 kJ/mol
H(298K)£½+ 49.4 kJ/mol
Ņ»¶ØĢõ¼žĻĀ£¬ĻņČŻ»żĪŖ2LµÄŗćČŻĆܱÕČŻĘ÷ÖŠ³äČė1 mol CH3OH(g)ŗĶ3 mol H2O(g)£¬ŹµŃé²āµĆ£ŗ“ļµ½Ę½ŗāדĢ¬Ź±£¬ĪüŹÕČČĮæ19.76 kJ”£Ōņ
¢Ł“ļĘ½ŗāŹ±»ģŗĻĘųĢåµÄŃ¹ĒæŹĒ·“Ó¦Ē°µÄ ±¶”£
¢ŚøĆĢõ¼žĻĀµÄøĆ·“Ó¦µÄĘ½ŗā³£ŹżŹĒ £Ø½į¹ū±£ĮōĮ½Ī»ÓŠŠ§Źż×Ö£©”£
¢ŪøĆĢõ¼žĻĀ·“Ó¦“ļĘ½ŗāדĢ¬µÄŅĄ¾ŻŹĒ£ØĢīŠņŗÅ£© ”£
| A£®vÕż(CH3OH)£½vÕż(CO2) | B£®»ģŗĻĘųĢåµÄĆÜ¶Č²»±ä |
| C£®c(CH3OH)£½c(H2O) | D£®»ģŗĻĘųĢåµÄ×ÜĪļÖŹµÄĮæ²»±ä |
 2SO3(g)
2SO3(g) 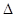 H£¼0£¬¼×”¢ŅŅĘšŹ¼·“Ó¦ĪĀ¶ČĻąĶ¬£¬¾łŗĶĶā½ēĪŽČČĮæ½»»»£¬Ę½ŗāŹ±£¬¼×ÖŠSO2µÄ×Ŗ»ÆĀŹĪŖa£¬ŅŅÖŠSO3µÄ·Ö½āĀŹĪŖb£¬Ōņa”¢bµÄ¹ŲĻµĪŖa+b _______ 1£ØĢī”°©”±”¢”°©”±»ņ”°£½”±) ”£
H£¼0£¬¼×”¢ŅŅĘšŹ¼·“Ó¦ĪĀ¶ČĻąĶ¬£¬¾łŗĶĶā½ēĪŽČČĮæ½»»»£¬Ę½ŗāŹ±£¬¼×ÖŠSO2µÄ×Ŗ»ÆĀŹĪŖa£¬ŅŅÖŠSO3µÄ·Ö½āĀŹĪŖb£¬Ōņa”¢bµÄ¹ŲĻµĪŖa+b _______ 1£ØĢī”°©”±”¢”°©”±»ņ”°£½”±) ”£²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø12·Ö£©æĘѧ¼ŅŅ»Ö±ÖĀĮ¦ÓŚ”°ČĖ¹¤¹ĢµŖ”±µÄ·½·ØŃŠ¾æ”£
£Ø1£©ŗĻ³É°±µÄŌĄķĪŖ£ŗN2(g)+3H2(g)  2NH3(g)
2NH3(g)  H="-92.4" kJ£Æmol£¬øĆ·“Ó¦µÄÄÜĮæ±ä»ÆČēĶ¼ĖłŹ¾”£
H="-92.4" kJ£Æmol£¬øĆ·“Ó¦µÄÄÜĮæ±ä»ÆČēĶ¼ĖłŹ¾”£
¢ŁŌŚ·“Ó¦ĢåĻµÖŠ¼ÓČė“߻ƼĮ£¬·“Ó¦ĖŁĀŹŌö“ó£¬E2µÄ±ä»ÆŹĒ (Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£
¢Ś½«0.3 mol N2ŗĶ0.5 mol H2³äČėĢå»ż²»±äµÄĆܱÕČŻĘ÷ÖŠ£¬ŌŚŅ»¶ØĢõ¼žĻĀ“ļµ½Ę½ŗā£¬²āµĆČŻĘ÷ÄŚĘųĢåŃ¹Ēæ±äĪŖŌĄ“µÄ £¬“ĖŹ±H2µÄ×Ŗ»ÆĀŹĪŖ £Ø¼ĘĖć³ö½į¹ū£©£»ÓūĢįøßøĆČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹ£¬ĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ (ĢīŃ”Ļī×ÖÄø)”£
£¬“ĖŹ±H2µÄ×Ŗ»ÆĀŹĪŖ £Ø¼ĘĖć³ö½į¹ū£©£»ÓūĢįøßøĆČŻĘ÷ÖŠH2µÄ×Ŗ»ÆĀŹ£¬ĻĀĮŠ“ėŹ©æÉŠŠµÄŹĒ (ĢīŃ”Ļī×ÖÄø)”£
| A£®ĻņČŻĘ÷ÖŠ°“Ō±ČĄżŌŁ³äČėŌĮĻĘų | B£®ĻņČŻĘ÷ÖŠŌŁ³äČėŅ»¶ØĮæH2 |
| C£®øı䷓ӦµÄ“߻ƼĮ | D£®Ņŗ»ÆÉś³ÉĪļ·ÖĄė³ö°± |
 4NH3(g)+3O2(g)
4NH3(g)+3O2(g)  H="+1530" kJ£Æmol
H="+1530" kJ£Æmol H="+44.0" kJ£Æmol
H="+44.0" kJ£Æmol 4NH3(g)+3O2(g)
4NH3(g)+3O2(g)  H = kJ£Æmol£¬øĆ·“Ó¦µÄ»ÆŃ§Ę½ŗā³£Źż±ķ“ļŹ½ĪŖK= £¬æŲÖĘĘäĖūĢõ¼ž²»±ä£¬Ōö“óŃ¹Ē棬KÖµ (Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£
H = kJ£Æmol£¬øĆ·“Ó¦µÄ»ÆŃ§Ę½ŗā³£Źż±ķ“ļŹ½ĪŖK= £¬æŲÖĘĘäĖūĢõ¼ž²»±ä£¬Ōö“óŃ¹Ē棬KÖµ (Ģī”°Ōö“ó”±”¢”°¼õŠ””±»ņ”°²»±ä”±)”£²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(14·Ö)¼Ó“ó¶ŌĆŗČ¼ÉÕ²śÉśµÄ·ĻĘų”¢·ĻŌüµÄ“¦ĄķŅŃæĢ²»ČŻ»ŗ”£
£Ø1£© ¶ŌČ¼ĆŗµÄ·ĻĘų½ųŠŠĶŃĻõ“¦ĄķŹ±£¬æÉĄūÓĆ¼×Ķé“߻ƻ¹ŌµŖŃõ»ÆĪļ£¬Čē£ŗ
CH4(g)£«4NO2(g)===4NO(g)£«CO2(g)£«2H2O(g)£»¦¤H£½a kJ”¤mol£1
CH4(g)£«4NO(g)===2N2(g)£«CO2(g)£«2H2O(g)£»¦¤H£½b kJ”¤mol£1
Ōņ·“Ó¦CH4(g)£«2NO2(g)===N2(g)£«CO2(g)£«2H2O(g)µÄ¦¤H£½________ kJ”¤mol£1(ÓĆŗ¬a”¢bµÄ“śŹżŹ½±ķŹ¾)”£
£Ø2£© ½«Č¼Ćŗ·ĻĘųÖŠµÄCO2×Ŗ»ÆĪŖ¶ž¼×Ćѵķ“Ó¦ŌĄķĪŖ£ŗ
2CO2(g)£«6H2(g)  CH3OCH3(g)£«3H2O(g)£»¦¤H
CH3OCH3(g)£«3H2O(g)£»¦¤H
¢Ł øĆ·“Ó¦Ę½ŗā³£Źż±ķ“ļŹ½ĪŖK£½________”£
¢Ś ŅŃÖŖŌŚÄ³Ń¹ĒæĻĀ£¬øĆ·“Ó¦ŌŚ²»Ķ¬ĪĀ¶Č”¢²»Ķ¬Ķ¶ĮĻ±ČŹ±£¬CO2µÄ×Ŗ»ÆĀŹČēÓŅĶ¼ĖłŹ¾”£øĆ·“Ó¦µÄ¦¤H________(Ģī”°>”±”°<”±»ņ”°£½”±)0”£ČōĪĀ¶Č²»±ä£¬¼õŠ”·“Ó¦Ķ¶ĮĻ±Č[n(H2) /n(CO2)]£¬ŌņK½«________(Ģī”°Ōö“ó”±”°¼õŠ””±»ņ”°²»±ä”±)”£
¢Ū ¶ž¼×ĆŃČ¼ĮĻµē³Ų¾ßÓŠĘō¶Ææģ£¬Š§ĀŹøßµČÓÅµć£¬Čōµē½āÖŹĪŖĖįŠŌ£¬¶ž¼×ĆŃČ¼ĮĻµē³ŲµÄøŗ¼«·“Ó¦ĪŖ____________________________”£
£Ø3£© ŅŌCO2ĪŖŌĮĻŗĻ³ÉµÄĢ¼Ėįõ„(½öŗ¬Ģ¼”¢Ēā”¢ŃõČżÖÖŌŖĖŲ)ŹĒÓĆĶ¾¹ć·ŗµÄ»Æѧʷ£¬Ä³ÖÖĢ¼Ėįõ„(DPC)Ė®½ā²śĪļÖ®Ņ»ÓöĀČ»ÆĢśČÜŅŗĻŌ×ĻÉ«£¬ÓŅĶ¼ĪŖĖüµÄŗĖ“Ź²ÕńĒāĘ×Ķ¼£¬ĒėŠ“³öDPCµÄ½į¹¹¼ņŹ½£ŗ____________”£
£Ø4£© ijµē³§µÄ·ŪĆŗ»Ņŗ¬ÄŖĄ“ŹÆ(Ö÷ŅŖ³É·ÖĪŖAl6Si2O13”¢SiO2)”£½«ĘäŗĶ“æ¼īŌŚøßĪĀĻĀÉÕ½į£¬æÉÖĘČ”NaAlSiO4(Ļ¼ŹÆ)”¢Na2SiO3ŗĶNaAlO2£¬ÓŠ¹Ų»Æѧ·½³ĢŹ½(·“Ó¦Ģõ¼ž¾łĪŖøßĪĀ)ĪŖ£ŗ
Al6Si2O13£«3Na2CO3===2NaAlSiO4£«4NaAlO2£«3CO2”ü
Al6Si2O13£«5Na2CO3===2Na2SiO3£«6NaAlO2£«5CO2”ü
SiO2£«Na2CO3===Na2SiO3£«CO2”ü
ŌņÓĆ1 mol Al6Si2O13ŗĶ4 mol SiO2ĶعżŅŌÉĻ·“Ó¦ÖʵĆ5 mol NaAlO2£¬¹²ĻūŗÄNa2CO3ĪŖ________mol”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
(15·Ö)Ģś¼°Ęä»ÆŗĻĪļÓėÉś²ś”¢Éś»ī¹ŲĻµĆÜĒŠ”£
£Ø1£©ĻĀĶ¼ŹĒŹµŃéŹŅŃŠ¾æŗ£Ė®¶ŌĢśÕ¢²»Ķ¬²æĪ»øÆŹ“ĒéæöµÄĘŹĆęŹ¾ŅāĶ¼”£
¢ŁøƵē»ÆøÆŹ“³ĘĪŖ ”£
¢ŚĶ¼ÖŠA”¢B”¢C”¢DĖÄøöĒųÓņ£¬Éś³ÉĢśŠā×ī¶ąµÄŹĒ (Ģī×ÖÄø)”£
£Ø2£©ÓĆ·ĻĢśĘ¤ÖĘČ”Ģśŗģ(Fe2O3)µÄ²æ·ÖĮ÷³ĢŹ¾ŅāĶ¼ČēĻĀ£ŗ
¢Ł²½ÖčIČōĪĀ¶Č¹żøߣ¬½«µ¼ÖĀĻõĖį·Ö½ā”£ĻõĖį·Ö½āµÄ»Æѧ·½³ĢŹ½ĪŖ ”£
¢Ś²½Öč¢ņÖŠ·¢Éś·“Ó¦£ŗ4Fe(NO3)2£«O2£«(2n£«4)H2O£½2Fe2O3”¤nH2O£«8HNO3£¬·“Ó¦²śÉśµÄHNO3ÓÖ½«·ĻĢśĘ¤ÖŠµÄĢś×Ŗ»ÆĪŖFe(NO3)2£¬øĆ·“Ó¦µÄ»Æѧ·½³ĢŹ½ĪŖ ”£
¢ŪÉĻŹöÉś²śĮ÷³ĢÖŠ£¬ÄÜĢåĻÖ”°ĀĢÉ«»Æѧ”±Ė¼ĻėµÄŹĒ (ČĪŠ“Ņ»Ļī)”£
£Ø3£©ŅŃÖŖt”ꏱ£¬·“Ó¦FeO(s)£«CO(g) Fe(s)£«CO2(g)µÄĘ½ŗā³£ŹżK£½0.25”£
Fe(s)£«CO2(g)µÄĘ½ŗā³£ŹżK£½0.25”£
¢Łt”ꏱ£¬·“Ó¦“ļµ½Ę½ŗāŹ±n(CO)£ŗn(CO2)£½ ”£
¢ŚČōŌŚ1 LĆܱÕČŻĘ÷ÖŠ¼ÓČė0.02 mol FeO(s)£¬²¢ĶØČėxmolCO£¬t”ꏱ·“Ó¦“ļµ½Ę½ŗā”£“ĖŹ±FeO(s)×Ŗ»ÆĀŹĪŖ50%£¬Ōņx£½ ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø17·Ö£©H2O2ŹĒŅ»ÖÖĒæŃõ»Æ¼Į£¬±»¹ć·ŗÓ¦ÓĆÓŚĖ®“¦Ąķ¼°ĪĄÉśĻū¶¾µČ·½Ćę”£
£Ø1£©H2O2²»ĪČ¶Ø£¬µ±ĘäÖŠŗ¬Fe2£«Ź±£¬»į·¢Éś·“Ó¦£ŗ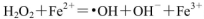
 £¬ŌņFe2£«ŌŚ“Ė¹ż³ĢÖŠĖłĘšµÄ×÷ÓĆŹĒ______________£¬µ±Éś³É336mL O2£Ø±ź×¼×“æö£©Ź±£¬·“Ó¦ÖŠ×ŖŅʵē×ÓµÄĪļÖŹµÄĮæĪŖ_______mol”£
£¬ŌņFe2£«ŌŚ“Ė¹ż³ĢÖŠĖłĘšµÄ×÷ÓĆŹĒ______________£¬µ±Éś³É336mL O2£Ø±ź×¼×“æö£©Ź±£¬·“Ó¦ÖŠ×ŖŅʵē×ÓµÄĪļÖŹµÄĮæĪŖ_______mol”£
£Ø2£©ĻĀ±ķŹĒŌŚ³£Ń¹”¢60”ęŗĶ²»Ķ¬pHĢõ¼žĻĀ£¬6mL30% H2O2ŌŚ60minÄŚŹĶ·Å³öŃõĘųµÄĢå»ż”£ŌņĻĀĮŠĖµ·ØÕżČ·µÄŹĒ___________”£
a£®pHŌ½“ó£¬H2O2µÄ·Ö½āĖŁĀŹŌ½“ó
b£®pHŌŚ9×óÓŅ£¬H2O2µÄ·Ö½āĖŁĀŹ×ī“ó
c£®6mL 30% H2O2·Ö½ā×ī¶ąŹĶ·Å³öµÄŃõĘųµÄĢå»żĪŖ639mL
d. pH=5.50Ź±£¬0”«60minÄŚ£¬v(O2)=1.5mL
£Ø3£©ČÜŅŗÖŠH2O2µÄ²ŠĮōĮææÉÓĆŅ»¶ØÅØ¶ČµÄĖįŠŌKMnO4ČÜŅŗĄ“²ā¶Ø£¬·“Ó¦ÖŠMnO4£
±»»¹ŌĪŖMn2£«£¬øĆ·“Ó¦µÄĄė×Ó·½³ĢŹ½ĪŖ______________”£
£Ø4£©æĘѧ¹¤×÷ÕßŅŌIr-Ru/TiĪŖŃō¼«”¢ACFCĪŖŅõ¼«£¬ŌŚĖįŠŌ»·¾³”¢²»¶ĻĶØČėæÕĘųµÄĢõ¼žĻĀÖ±½Óµē½āĖ®Ą“ÖʱøH2O2”£µē½ā¹ż³ĢÖŠ£¬Ńō¼«ĒųČÜŅŗµÄpH_ £ØĢī”°Ōö“ó”±”°²»±ä”±»ņ”°¼õŠ””±£©£¬Ņõ¼«²śÉśH2O2µÄµē¼«·“Ó¦Ź½ĪŖ_______”£Čō²»ĶØæÕĘų£¬ŌņŅõ¼«µĆµ½µÄ²śĪļŹĒ_______ ”£
£Ø5£©¼ŗÖŖ¶ĻĮŃ1mol»Æѧ¼üĖłŠčµÄÄÜĮæ
143,H-OĪŖ463”£Ōņ .
.
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
ĮņĖįµÄĻū·ŃĮæŹĒŗāĮæŅ»øö¹ś¼Ņ»Æ¹¤Éś²śĖ®Ę½µÄÖŲŅŖ±źÖ¾”£
£Ø1£©Ņ»¶ØĢõ¼žĻĀ£¬SO2ÓėæÕĘų·“Ó¦10 minŗó£¬SO2ŗĶSO3ĪļÖŹµÄĮæÅØ¶Č·Ö±šĪŖ1.2 mol/LŗĶ2.0 mol/L£¬ŌņSO2ĘšŹ¼ĪļÖŹµÄĮæÅضČĪŖ______£»Éś³ÉSO3µÄ»Æѧ·“Ó¦ĖŁĀŹĪŖ______”£
£Ø2£©ČōøıäĶā½ēĢõ¼ž£¬·¢ÉśSO2ÓėæÕĘų·“Ӧɜ³ÉSO3£¬Ź¹10 minÄŚµÄÓĆO2±ķŹ¾µÄ·“Ó¦ĖŁĀŹĪŖ0.15mol/(L”¤min)£¬ŌņøıäµÄĢõ¼žæÉÄÜŹĒ_______________”£
| A£®Ń¹ĖõĢå»ż£¬Ōö“óŃ¹Ēæ | B£®½µµĶĪĀ¶Č | C£®³äČė“óĮæµÄµŖĘų | D£®½öŌö¼ÓSO2µÄÅØ¶Č |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
£Ø10·Ö£¬ĆææÕ2·Ö£© ĶłŅ»øöČŻ»żĪŖ5LµÄĆܱÕČŻĘ÷ÖŠ£¬ĶØČė2molN2ŗĶ3molH2£¬ŌŚŅ»¶ØĢõ¼žĻĀ,·¢Éś·“Ó¦ N2+3H2 2NH3£¬2 minŗó²āµĆČŻĘ÷ÖŠÉś³ÉĮĖ1mol NH3£¬ŹŌĒó£ŗ
2NH3£¬2 minŗó²āµĆČŻĘ÷ÖŠÉś³ÉĮĖ1mol NH3£¬ŹŌĒó£ŗ
£Ø1£©2 minŹ±£¬N2µÄĪļÖŹµÄĮæ £» £Ø2£©2 minŹ±£¬H2µÄĪļÖŹµÄĮæÅØ¶Č £»
£Ø3£©2 minŹ±£¬H2µÄ×Ŗ»ÆĀŹ £» £Ø4£©2minÄŚ£¬N2µÄĘ½¾ł·“Ó¦ĖŁĀŹ £»
£Ø5£©N2”¢H2”¢NH3µÄ·“Ó¦ĖŁĀŹÖ®±Č ”£
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ ĢāŠĶ£ŗĢīæÕĢā
·“Ó¦A(g)  B(g) +C(g)ŌŚČŻ»żĪŖ1.0LµÄĆܱÕČŻĘ÷ÖŠ½ųŠŠ£¬AµÄ³õŹ¼ÅضČĪŖ0.050mol/L”£ĪĀ¶ČT1ŗĶT2ĻĀAµÄÅضČÓėŹ±¼ä¹ŲĻµČēĶ¼ĖłŹ¾”£»Ų“šĻĀĮŠĪŹĢā£ŗ
B(g) +C(g)ŌŚČŻ»żĪŖ1.0LµÄĆܱÕČŻĘ÷ÖŠ½ųŠŠ£¬AµÄ³õŹ¼ÅضČĪŖ0.050mol/L”£ĪĀ¶ČT1ŗĶT2ĻĀAµÄÅضČÓėŹ±¼ä¹ŲĻµČēĶ¼ĖłŹ¾”£»Ų“šĻĀĮŠĪŹĢā£ŗ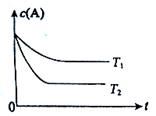
£Ø1£©ÉĻŹö·“Ó¦µÄĪĀ¶ČT1 T2£¬Ę½ŗā³£ŹżK£ØT1£© K£ØT2£©”££ØĢī”°“óÓŚ”±”¢”°Š”ÓŚ”±»ņ”°µČÓŚ”±£©
£Ø2£©ČōĪĀ¶ČT2Ź±£¬5minŗó·“Ó¦“ļµ½Ę½ŗā£¬AµÄ×Ŗ»ÆĀŹĪŖ70%£¬Ōņ£ŗ
¢ŁĘ½ŗāŹ±ĢåĻµ×ܵÄĪļÖŹµÄĮæĪŖ ”£
¢Ś·“Ó¦µÄĘ½ŗā³£ŹżK= ”£
¢Ū·“Ó¦ŌŚ0~5minĒų¼äµÄĘ½¾ł·“Ó¦ĖŁĀŹv(A)= ”£
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com