
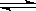 CH3OH(g)��ͼI��ͼ���ǹ��ڸ÷�Ӧ���������ͼʾ��
CH3OH(g)��ͼI��ͼ���ǹ��ڸ÷�Ӧ���������ͼʾ��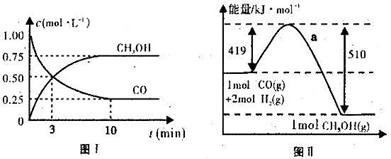
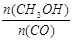 ������� ��
������� ��| ���� | �� | �� | �� |
| Ͷ���� | 1mol CO ��2mol H2 | 1molCH3OH | 2molCH3OH |
| CH3OH��Ũ�ȣ�mol��L-1�� | c1 | c2 | c3 |
| ��Ӧ�������仯 | �ų�Q1kJ | ����Q2kJ | ����Q3kJ |
| ��ϵѹǿ��Pa�� | P1 | P2 | P3 |
| ��Ӧ��ת���� | ��1 | ��2 | ��3 |
 E��2��2=��3
E��2��2=��3
 CH3OH(g)����H= -91kJ��mol-1
CH3OH(g)����H= -91kJ��mol-1 ��С
��С 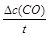 =
= =0.075mol/(L��min)��
=0.075mol/(L��min)��
 ����ӦΪ���ȷ�Ӧ�������¶ȣ�ƽ�������ƶ���K��С��
����ӦΪ���ȷ�Ӧ�������¶ȣ�ƽ�������ƶ���K��С�� ��С������He����ʹ�ô�����ƽ�ⲻ�ƶ���
��С������He����ʹ�ô�����ƽ�ⲻ�ƶ��� ���䣻�ٳ���1molCO��2molH2��Ч������ѹǿ��ƽ�������ƶ���
���䣻�ٳ���1molCO��2molH2��Ч������ѹǿ��ƽ�������ƶ��� ����
����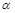 1+
1+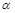 2=1��Q1+Q2=91��p1=p2��
2=1��Q1+Q2=91��p1=p2��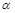 3<
3<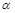 2��
2��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
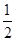 O2(g)===H2O(g)����H����241.8 kJ��mol��1������˵���в���ȷ���� �� ��
O2(g)===H2O(g)����H����241.8 kJ��mol��1������˵���в���ȷ���� �� ��| A��H2��ȼ����Ϊ241.8 kJ��mol��1 |
| B��2H2(g)��O2(g)===2H2O(g)����H����483.6 kJ��mol��1 |
| C��1 mol H2��ȫȼ������Һ̬ˮ�ų�����������241.8 kJ |
| D���Ͽ�1 mol H2O�Ļ�ѧ�����յ����������ڶ���1 mol H2��0.5 mol O2�Ļ�ѧ�������յ������� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��1 ��1 | B��2 ��1 | C��2 ��3 | D��1 ��3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����317.3 kJ/mol | B����379.3 kJ/mol | C����332.8 kJ/mol | D��317.3 kJ/mol |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ�������
| �� �� | ȼ���ȣ�kJ��mol��1�� |
| H2(g) | ��285.8 |
| CO(g) | ��283.0 |
| CH4(g) | ��890.3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
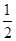 O2(g)=H2O(l) ��H=��285.84kJ��mol��1
O2(g)=H2O(l) ��H=��285.84kJ��mol��1 �鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������
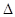 H=" -198" kJ��mol-1��Ӧ���̵������仯��ͼ��ʾ����ش��������⣺
H=" -198" kJ��mol-1��Ӧ���̵������仯��ͼ��ʾ����ش��������⣺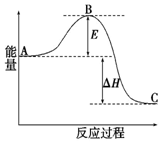
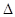 H= kJ��mol-1;
H= kJ��mol-1; �鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
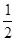 O2(g)==H2O(l) ��H3= ��285 kJ��mol-1
O2(g)==H2O(l) ��H3= ��285 kJ��mol-1| A����243.5 kJ��mol-1 | B����487 kJ��mol-1 | C����1113.5 kJ��mol-1 | D����2227 kJ��mol-1 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com