


 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ��Ķ�����
| �ζ����� ʵ������ |
1 | 2 | 3 | 4 |
| V��������Һ��/mL | 20.00 | 20.00 | 20.00 | 20.00 |
| V��NaOH��/mL���������� | 0.00 | 0.200 | 0.10 | 0.00 |
| V��NaOH��/mL���ն����� | 14.98 | 15.20 | 15.12 | 15.95 |

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2012���㽭ʡ����һ�и�����ѧ�ڻ�ͷ���Ի�ѧ�Ծ� ���ͣ�ʵ����
ʳ��Ϊ��������ƴ����֡����ұ��涨����ʳ�����Ậ�����õ���3��5g��100mL��
��1��ij�о�С�����õζ��������ⶨijƷ��ʳ���д���ĺ���������˵����ȷ����______
a����NaOH��Һ�ζ�ʱ��Ӧ�����ӷ���ʽΪ��H++OH-=H2O
b��ʳ��������ϡ��һ���������ٽ��еζ�
c����NaOH��Һ�ζ�ʳ�ף���ʹ�÷�̪�������ָʾ��
d������ø�Ʒ��ʳ�����ʵ���Ũ��Ϊ0.75mol��L-1�����ʳ��������Ϊ4.5g/100mL��
��2���о�С���ͬѧ��ϸ�۲��˸�Ʒ��ʳ�ı�ǩ���������л����б���������ΪʳƷ���Ӽ������������Ϸ���֤������ʳƷ���Ӽ�����������C6H5COONa�����ᷢ�����ӻ�����Ӧ���������һ���¶��µĴ����뱽�����________ ����д��ţ���
a��pH b������� c�����볣�� d���ܽ��
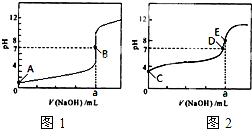
��3�������£���0.1000 mol��L-1NaOH��Һ�ֱ�ζ�20.00mL 0��1000 mol��L-1HCl��Һ��20.00mL 0.1000 mol��L-1CH3COOH��Һ���õ�2���ζ����ߣ�����ͼ��ʾ��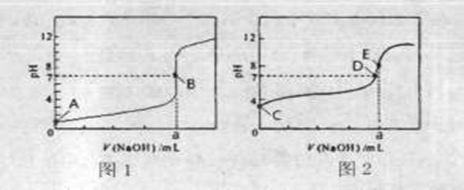
�ٵζ�������Һ��������_______ ���ͼl����ͼ2�������ζ�������a=_____ mL��
��E��pH>8��ԭ�������ӷ���ʽ��ʾ________��
��4���������Բⶨһδ֪Ũ�ȵ�CH3COOH��Һ�ĵ���ȣ�Ӧ����ʵ���������Լ�����ֽ����________����д��ţ�
a����⣬NaOH��Һ b������Na2CO3��Һ
c��������Ӧ��ʯ����Һ d���к͵ζ���pH��ֽ
��5��Ϊ�궨ij������Һ��ȷŨ�ȣ���0.2000 mol?L-1 ��NaOH��Һ��20.00 mL������Һ���еζ������εζ�����NaOH��Һ��������£�
| ʵ����� | 1 | 2 | 3 | 4 |
| ����NaOH��Һ�������mL�� | 20.05 | 20.00 | 18.80 | 19.95 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2013���㽭ʡ�߶���ѧ�����п��Ի�ѧ�Ծ��������棩 ���ͣ������
��10�֣�ʳ��Ϊ��������ƴ����֡����ұ��涨����ʳ�����Ậ�����õ���3.5g/100mL��
��1��ij�о�С�����õζ��������ⶨijƷ��ʳ���д���ĺ���������˵����ȷ���� ��
a.��NaOH��Һ�ζ�ʱ��Ӧ�����ӷ���ʽΪ��H++OH-=H2O
b.ʳ��������ϡ��һ���������ٽ��еζ�
c.��NaOH��Һ�ζ�ʳ�ף���ʹ�÷�̪�������ָʾ��
d.����ø�Ʒ��ʳ�����ʵ���Ũ��Ϊ0.75mol��L-1�����ʳ��������Ϊ4.5g/100mL��
��2���о�С���ͬѧ��ϸ�۲��˸�Ʒ��ʳ�ı�ǩ���������л����б���������ΪʳƷ���Ӽ������������Ϸ���֤������ʳƷ���Ӽ�����������C6H5COONa�����ᷢ�����ӻ�����Ӧ���������һ���¶��µĴ����뱽����� ����д��ţ���
a.pH b.����� c.���볣�� d.�ܽ��
��3�������£���0.1000 mol��L-1NaOH��Һ�ֱ�ζ�20.00mL 0.1000 mol��L-1HCl��Һ��20.00mL 0.1000 mol��L-1CH3COOH��Һ���õ�2���ζ����ߣ�����ͼ��ʾ��
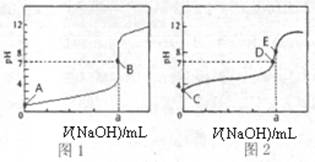
�ٵζ�������Һ�������� ���ͼl����ͼ2�������ζ�������a= mL��
��E��pH>8��ԭ�������ӷ���ʽ��ʾ ��
��4���������Բⶨһδ֪Ũ�ȵ�CH3COOH��Һ�ĵ���ȣ�Ӧ����ʵ���������Լ�����ֽ���� ��
a.��⣬NaOH��Һ b.����Na2CO3��Һ
c.������Ӧ��ʯ����Һ d.�к͵ζ���pH��ֽ
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com