

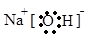
 2NH3
2NH3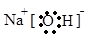 ���� H�Ļ�ѧʽΪ NaAlO2��
���� H�Ļ�ѧʽΪ NaAlO2�� 2NH3 ���� д����Ӧ�ݵ����ӷ���ʽAl3++3NH3+3H2O=Al(OH)3��+3NH4+ �� Al3++3NH3��H2O=Al(OH)3��+3NH4+��
2NH3 ���� д����Ӧ�ݵ����ӷ���ʽAl3++3NH3+3H2O=Al(OH)3��+3NH4+ �� Al3++3NH3��H2O=Al(OH)3��+3NH4+��


| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A���� | B��̼ | C���� | D���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A����A | B����B | C����A | D����B |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ������

�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
| A��ԭ�Ӱ뾶��C<Be<Mg<Na |
| B���ȶ��ԣ�HI>HBr>HCl>HF |
| C�����ԣ�H4SiO4<H3PO4<H2SO4<HClO4 |
| D�����ԣ�KOH>Ca(OH)2>Mg(OH)2>Al(OH)3 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ������ ���ͣ���ѡ��
 �������й����ʵ��ƶϲ���ȷ����
�������й����ʵ��ƶϲ���ȷ����| A������Ϊ��̿��������O2 |
| B������ΪSO2�������ǰ�ˮ |
| C������ΪFe������������ |
| D������ΪNaOH��Һ��������CO2 |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com