
�Ӻ�ˮ��ȡþ������������£���ش�������⡣
��1���Ӻ�ˮ����ȡþ����������ͼ��ʾ����ͼ������Ҫ�����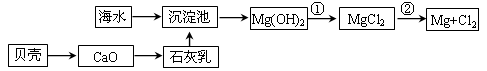
i.��ˮ���������ǰ���Ժ�ˮ���д������������ַ�����
����һ����ɹ�κ��±ˮͨ������أ�
������������������Ũ����ĺ�ˮͨ������ء�
��������________��������������_________________________________________��
ii.��Ӧ�ٵ����ӷ�����________________________________________________��
��Ӧ�ڵĻ�ѧ����ʽ��_______________________________________________��
��2���Ӻ�ˮ����ȡ�����������ͼ��ʾ����ͼ������Ҫ�����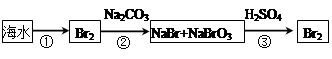

i.���̢��У�������Լ���___________��
ii.���̢��У�����Һ�д����ȿ��������崵�����ô������գ������ȿ�����Ŀ����______________________________________________________________________��
iii.���̢��з�Ӧ�Ļ�ѧ����ʽ��____________________________________________��
iv.�����յõ����嵥������Ȼ����������Cl2�����ȥ�����ʵķ�����__________________________________________________��������ӷ���ʽ�ش𣩡�
��9�֣���1��i. һ Ҫ���Ⱥ�ˮ���������Դ���˷�����Դ
ii. Mg(OH)2��2H����Mg2����2H2O �� MgCl2(����) Mg��Cl2��
Mg��Cl2��
��2��i. Cl2
ii.�¶����ߣ�������ܽ�ȼ�С��������������巢����Ӧ�����Խ���Һ�壬���������������ݳ�
iii.5NaBr��NaBrO3��3H2SO4��3Br2��3Na2SO4��3H2O��2�֣�
iv.���������廯�ƣ������л��ܼ���������ᴿ��Cl2��2Br����Br2��2Cl��
���������������1��i.��������Ũ����ˮ��Ҫ���Ⱥ�ˮ���Ӷ����Ĵ�������Դ���˷�����Դ�����Է���һ��á�
ii.������þת��Ϊ�Ȼ�þ����Ҫ��������þ�ܽ��������У���Ӧ�����ӷ���ʽ��Mg(OH)2��2H����Mg2����2H2O��þ�ǻ��õĽ�������Ҫ��ⷨұ����������ڵ��Ȼ�þ�������ɽ���þ����������Ӧ�Ļ�ѧ����ʽ��MgCl2(����) Mg��Cl2����
Mg��Cl2����
��2��i.��ˮ�е������������ӵ��γɴ��ڵģ���Ҫ���������������������ɵ����壬��˹��̢��У�������Լ���Cl2��
ii.�����¶����ߣ�������ܽ�ȼ�С��������������巢����Ӧ�����Խ���Һ�壬���������������ݳ������Ҫ�����ȿ�����
iii.�����������£��������ܺ��廯�Ʒ���������ԭ��Ӧ���ɵ����壬��Ӧ�Ļ�ѧ����ʽ��5NaBr��NaBrO3��3H2SO4=3Br2��3Na2SO4��3H2O��
iv. ��������ǿ�����ԣ�����Ҫ��ȥ�������е��������������Լ��������廯�ƣ������л��ܼ���������ᴿ����Ӧ�����ӷ���ʽ��Cl2��2Br��=Br2��2Cl����
���㣺���麣ˮ�н���þ�������ȡ��ʵ�鷽�����ۺͷ���ʽ����д
�����������Ǹ߿��еij������ͣ������е��Ѷȵ����⡣�����ۺ���ǿ�����ض�ѧ�������������ͽ��ⷽ����ָ����ѵ����ּ�ڿ���ѧ��������û���֪ʶ���ʵ�����������������������ѧ����Ӧ�������������������������Ժ�ˮ���ۺ�Ӧ��Ϊ���壬�����ڵ���ѧ����ѧϰ��Ȥ������ѧ����ѧϰ��֪����


 ���ɶ��ȫ���ƿؾ�ϵ�д�
���ɶ��ȫ���ƿؾ�ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
�йغ����в�����Դ�����ã���ͼ����˵������ȷ����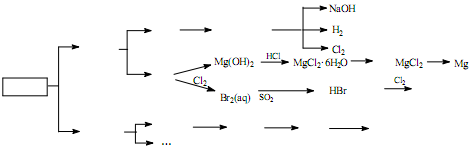
| A����ҵ������Cl2�����ʯ��ˮ��Ӧ��ȡƯ�� |
| B����MgCl2��6H2O��������ˮMgCl2������HCl�����м�����ˮ |
| C��SO2ˮ��Һ����Br2�Ļ�ѧ����ʽΪ��Br2+SO2+2H2O=H2SO4+2HBr |
| D���ֵ��л�������HCl��IBr�ɼ���KI���������ᴿ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����������һ����;ʮ�ֹ㷺�Ļ���ԭ�ϡ���ҵ����Ҫͨ������Ȼ��Ʊ�����Һ�ķ�������������ƣ��ҹ����ȼҵ������������ӽ���Ĥ���ۡ�
(1)���ӽ���Ĥ����һ����ý���������������ԭ����__________________________________��
����һ����̼�����Ƴɡ������ӽ���Ĥ�ѵ��۸��������Һ������ң���������____________________________________��
(2)Ϊʹ����Ȼ��Ƶ����ʼӿ죬���д�ʩ���е���__________��
a����������̼���������
b����߱����Ȼ�����Һ���¶�
c���Ӵ�������������ľ���
d����ߵ��ʱ��Դ��ѹ
(3)�����ij���ӽ���Ĥ���۵Ģٵ��ʱ��ѹ����ԭ����2�����ڵ��ʱ�ĵ���ǿ������ԭ����2�����۵��ʱ�¶ȴ�30 ����ߵ�60 �棬��������һ����ﵽԭ����2������________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������Դ�ı��⣬��ˮ�м����������е���ȻԪ�ء�������Դ��ѧ�����о��Ӻ�������ȡ��ѧ���ʵ�ѧ�ƣ������о��Ӻ�������ȡ����Ԫ���⣬���о��Ӻ�������ȡ��Ԫ�أ�Ũ��С��1mg/L����
��1�������к�����ߵ�±��Ԫ�������ڱ��е�λ��Ϊ ������ͬ���������ҵ���Ϊ�����Ԫ��ԭ�ӵĺ�������Ų�ʽΪ ��
��2��������Ԫ�غ���λ��ǰ�е�Ԫ���������ȡ��ơ�þ���������Ӱ뾶�Ӵ�С��˳��Ϊ �������ӷ��ű�ʾ���������γɵĻ��������ܷ���������� ���õ���ʽ��ʾ����
��3����Ԫ�����ں�ˮ����Ҫ��Be(OH)+��ʽ���ڣ�����������Ԫ�����ƣ�Ŀǰ�Ǵ��̱�ʯ����Ҫ�ɷ�Ϊ����������Be3Al2Si6O18������ȡ���������Ǻ��ա����ӡ������ȹ�ҵ���������ս�Խ������ϣ���˺�ˮ������Ϊ������Դ��ѧ�µ��о�������д����
�����������ε���������ʽ�Ļ�ѧʽ�� ��
��Be(OH)+��ǿ����Һ��Ӧ�����ӷ���ʽ�� ��
��4�����дӺ�������ȡ���ᴿ���ʵ�������ʵ�������У����������� ��ѡ���ţ���
a����ˮ���壺��ˮŨ��
 ������
������

 Һ��
Һ��
b����ˮ��þ����̲����
 ʯ����
ʯ����

 MgO
MgO þ
þ
c��������⣺��������
 ��Һ
��Һ �����л���Һ
�����л���Һ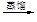 �⾧��
�⾧��
d�������ᴿ������

 ����
����

 ��Һ
��Һ
 ʳ�ξ���
ʳ�ξ���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ���������������ͼ���£���ش��������⣺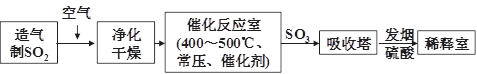
��1���������������Ի�����Ϊԭ�ϣ������ڹ����������������Ϊԭ�ϣ������� ��
��2��������������Ӧ��ǰ�辻����ԭ���� ��
��3���ڴ���Ӧ����ͨ��ʹ�ó�ѹ���ڴ�������SO2��ת����Ϊ90%�����Dz��ַ�����Ҳ�ȡ��ѹ��������ȡSO3����ȡ��ѹ��ʩ��Ŀ�ij��˼ӿ췴Ӧ�����⣬������ ���Ӷ��������Ч�ʡ�
��4����ҵ�����г��ð����ᷨ����β�������Դﵽ������Ⱦ���������õ�Ŀ�ġ��û�ѧ����ʽ��ʾ�䷴Ӧԭ���� ��
��5�������Ṥҵ�⣬�������ҵ������������صĹ�ҵ������������ȷ���� ��
A����ˮ���壺��ˮŨ��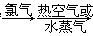 ������ ������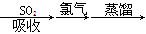 Һ�� Һ�� |
B����ˮ��þ����̲����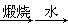 ʯ��ˮ ʯ��ˮ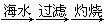 MgO MgO þ þ |
C����ҵ���������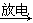 NO2 NO2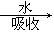 �����β������ �����β������ |
D����ҵ�ϳɰ�����Ȼ�� ���� ���� NH3��H2��N2 NH3��H2��N2 �� �� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
���й��ڻ�ѧ���������������ʶ�������
| A�������÷���֬�Ʒ��� |
| B��ˮ��������������ľ�ķ���� |
| C��ú��������Һ���������仯�ɱ�Ϊ���ȼ�� |
| D���������ƳɵIJ۳������ܷ�����Ũ�����Ũ���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
ﯲ�ҵ�Ǽ��з�չDZ����ǰ�������˲�ҵ,�(Zr)Ԫ���Ǻ˷�Ӧ��ȼ�ϰ��İ�������,�������(ZrO2)�����������������մɡ��ҹ��зḻ���Ӣʯ(ZrSiO4),��Al2O3��SiO2��Fe2O3������,���������֮һ����: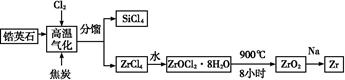
�Իش���������:
(1)д�����������и��������ķ�Ӧ����ʽ(̼ת����CO): ;
(2)д��ZrOCl2��8H2O��900 ������ZrO2�ķ�Ӧ����ʽ�� ;
(3)���ڶ�����������մɺ�ﯺϽ��˵������ȷ������������(��ѡ)��
| A��������������մ����������ǽ������� |
| B��1����=10-10�� |
| C��ﯺϽ��Ӳ�ȱȴ��Ҫ�� |
| D���ձ������˵�վ�ı�ը��������ﯺϽ��ڸ�������ˮ������Ӧ������������ը���� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��ҵ�����������ᣨ�е㣺90oC��ʱ��ͬʱ�������������ƣ��乤���������£�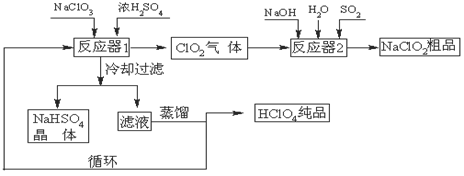
��1����ȴ���˵�Ŀ���ǽ���NaHSO4�� ���������NaHSO4���塣
��2����Ӧ��2�з�����Ӧ�����ӷ���ʽΪ ��SO2��
�������� ����
��3��������ҵ����������Ļ�ѧ��ӦΪ:3NaClO3+3H2SO4��Ũ����3NaHSO4+HClO4+2ClO2+H2O�����������뻹ԭ��������ʵ���֮��Ϊ ��
��4������ͨ��������Һ�ķ����õ��������ԭ������Ǹ�����ķе�Ƚ� ����ߡ��͡��������״���Һ���ݳ���ѭ��ʹ�õ������� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ���ѡ��
ij���ϲ��������˹�̼��άΪ��ǿ�塢������Ϊ���帴�϶��ɵġ��������ֲ��Ͼ��е����ʻ���;��(����)��
�����£��ڲ����ȣ��۵��硢���ȣ��ܲ����硢�����ȣ��ݿ����ڷɻ������������ڵ����Ŀ���
| A���٢ۢݢ� | B���ڢۢݢ� |
| C���ڢۢ� | D���ۢܢ� |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com