
 O2��g���TH2O��l����H3=-285.8kJ/mol
O2��g���TH2O��l����H3=-285.8kJ/mol H2SO4��aq��+NaOH��aq��=
H2SO4��aq��+NaOH��aq��= Na2SO4��aq��+H2O��l������H=-57.3kJ/mol��
Na2SO4��aq��+H2O��l������H=-57.3kJ/mol�� H2SO4��aq��+NaOH��aq��=
H2SO4��aq��+NaOH��aq��= Na2SO4��aq��+H2O��l������H=-57.3kJ/mol��
Na2SO4��aq��+H2O��l������H=-57.3kJ/mol�� =0.25mol��
=0.25mol��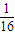 mol
mol 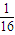 mol�Ķ���ų�������ΪQ�������ȼ����Ϊ16Q��
mol�Ķ���ų�������ΪQ�������ȼ����Ϊ16Q��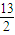 O2��g��=4CO2��g��+5H2O��l����H=-16QkJ/mol��
O2��g��=4CO2��g��+5H2O��l����H=-16QkJ/mol�� O2��g��=4CO2��g��+5H2O��l����H=-16QkJ/mol��
O2��g��=4CO2��g��+5H2O��l����H=-16QkJ/mol�� O2��g���TH2O��l ����H3=-285.8kJ/mol
O2��g���TH2O��l ����H3=-285.8kJ/mol

 �»����ܶ�Ա��ϵ�д�
�»����ܶ�Ա��ϵ�д� ����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
����ͼ����ּ��������ҵ֣�ݴ�ѧ������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
| 1 |
| 2 |
| 1 |
| 2 |
| 1 |
| 2 |
| 1 |
| 2 |
| 13 |
| 2 |
| 13 |
| 2 |
| 1 |
| 2 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
(6��)��Ҫ��д�Ȼ�ѧ����ʽ��
(1)��֪ϡ��Һ�У�1mol H2SO4��NaOH��Һǡ����ȫ��Ӧ��������ʱ���ų�114.6 kJ������д����ʾH2SO4��NaOH��Ӧ���Ȼ�ѧ����ʽ_____________________��
(2)25�桢101 kPa�����³��ȼ��һ�����Ķ�������ų�����ΪQ kJ�����ⶨ�������ɵ�CO2ͨ����������ʯ��ˮ�в���25 g��ɫ������д����ʾ����ȼ���ȵ��Ȼ�ѧ����ʽ ��
(3)��֪�����Ȼ�ѧ����ʽ��
��CH3COOH(l)��2O2(g)===2CO2(g)��2H2O(l) ��H1����870.3 kJ/mol
��C(s)��O2(g)===CO2(g) �� ��H2����393.5 kJ/mol
��H2(g)��1/2O2(g)===H2O(l) ��H3����285.8 kJ/mol
д����C(s)��H2(g)��O2(g)��������CH3COOH(l)���Ȼ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012ѧ��ɽ��ʡ��ͬһ�и߶���ѧ�����п��Ի�ѧ�Ծ� ���ͣ���ѡ��
(8��)��Ҫ��д�Ȼ�ѧ����ʽ��
��֪ϡ��Һ�У�1 mol H2SO4��NaOH��Һ
ǡ����ȫ��Ӧʱ���ų�114.6 kJ������
д����ʾH2SO4��NaOH��Ӧ���к��ȵ��Ȼ�ѧ����ʽ
_________________________________________________________________.
(2) 25�桢101 kPa�����³��ȼ��һ�����Ķ�������ų�����ΪQ kJ�����ⶨ�������ɵ�CO2ͨ����������ʯ��ˮ�в���25 g��ɫ������д����ʾ����ȼ���ȵ��Ȼ�ѧ����ʽ______________________________________________________��
(3) ��֪�����Ȼ�ѧ����ʽ��
��CH3COOH(l)��2O2(g)===2CO2(g)��2H2O(l) ��H1����870.3 kJ/mol
��C(s)��O2(g)===CO2(g)����H2����393.5 kJ/mol
��H2(g)��O2(g)===H2O(l)��H3����285.8 kJ/mol
д����C(s)��H2(g)��O2(g)��������CH3COOH(l)���Ȼ�ѧ����ʽ_________________________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��2011-2012���½���³ľ���еڰ���ѧ��һ��ѧ����ĩ���Ի�ѧ�� ���ͣ������
(9��)��Ҫ��д�Ȼ�ѧ����ʽ��
(1)��֪ϡ��Һ�У�1 mol H2SO4��NaOH��Һǡ����ȫ��Ӧʱ���ų�114.6 kJ������д����ʾH2SO4��NaOH��Ӧ���к��Ȼ�ѧ����ʽ______________________________________.
(2)25�桢101 kPa�����³��ȼ��һ�����Ķ�������ų�����ΪQ kJ�����ⶨ�������ɵ�CO2ͨ����������ʯ��ˮ�в���25 g��ɫ������д����ʾ����ȼ���Ȼ�ѧ����ʽ___________________��
(3)��֪�����Ȼ�ѧ����ʽ��
��CH3COOH(l)��2O2(g)===2CO2(g)��2H2O(l) ��H1����870.3 kJ/mol
��C(s)��O2(g)===CO2(g) �� ��H2����393.5 kJ/mol
��H2(g)��O2(g)===H2O(l ) ��H3����285.8 kJ/mol
д����C(s)��H2(g)��O2(g)��������CH3COOH(l)���Ȼ�ѧ����ʽ_______________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ��ɽ��ʡ2010�����5��ĩ�����������ۻ�ѧ ���ͣ������
(6��)��Ҫ��д�Ȼ�ѧ����ʽ��
(1)��֪ϡ��Һ�У�1 mol H2SO4��NaOH��Һǡ����ȫ��Ӧ��������ʱ���ų�114.6 kJ������д����ʾH2SO4��NaOH��Ӧ���Ȼ�ѧ����ʽ_____________________��
(2)25�桢101 kPa�����³��ȼ��һ�����Ķ�������ų�����ΪQ kJ�����ⶨ�������ɵ�CO2ͨ����������ʯ��ˮ�в���25 g��ɫ������д����ʾ����ȼ���ȵ��Ȼ�ѧ����ʽ ��
(3)��֪�����Ȼ�ѧ����ʽ��
��CH3COOH(l)��2O2(g)===2CO2(g)��2H2O(l) ��H1����870.3 kJ/mol
��C(s)��O2(g)===CO2(g) �� ��H2����393.5 kJ/mol
��H2(g)��1/2O2(g)===H2O(l ) ��H3����285.8 kJ/mol
д����C(s)��H2(g)��O2(g)��������CH3COOH(l)���Ȼ�ѧ����ʽ ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com