
����Ŀ�����仯�����ڹ�ũҵ����������Ҫ�����á�
(1)PԪ���а��ס����ס��������ֳ����ĵ��ʡ�
���ִ���ѧ�У�������_______�ϵ���������������Ԫ�ء�
�ڰ���(P4)������CS2��������ˮ��ԭ����_________��
�ۺ�����һ�ֺ�ɫ�н�������ľ��壬��һ�ֱ�ʯīϩ����������Ͳ��ϡ����ס������Ƿ��Ӿ��壬�����������ʯī�����ƵIJ�״�ṹ������ͼ��ʾ�������йغ������˵������ȷ����_______��
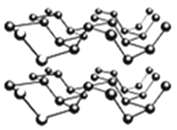
A������������ԭ���ӻ���ʽΪsp3�ӻ�
B���������в����֮����������Ƿ��Ӽ�������
C���������ÿһ������ԭ�Ӷ���ͬһƽ����
D��PԪ�����ֳ����ĵ����У������۷е����
(2)������һ����ĥͿ�ϣ��������������ı��汣���㡣����������廯�����廯���ڸ������������з�Ӧ�ϳɡ�������ľ�������ͼ��ʾ������ʵ����Ϊ��ԭ�ӡ�

��ش��������⣺
������Ļ�ѧʽΪ_________���þ���ľ���������___________��
�����廯������Ԫ�ػ�̬ԭ�ӵĵ����Ų�ʽΪ_________�����廯����ӵĿռ乹����________���ϳ�����Ļ�ѧ����ʽΪ��___________��
����һ����������ԭ�ӿռ�ѻ���ʽΪ________����ԭ�ӵ���λ��Ϊ________���ýṹ����һ����λ�����ṩ�չ����ԭ����_______����֪������B��Pԭ�ӵ��������Ϊa nm����þ�����ܶȵı���ʽΪ(���軯��)________g/cm3��
���𰸡�ԭ�ӹ��� P4��CS2�ǷǼ��Է��ӣ�H2O�Ǽ��Է��ӣ�������������ԭ����P4������ˮ C BP ԭ�Ӿ��� 1s22s22p63s23p63d104s24p5��[Ar] 3d104s24p5 ƽ�������� PBr3��BBr3��3H2![]() BP��6HBr �����������ܶѻ� 4 B
BP��6HBr �����������ܶѻ� 4 B 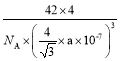
��������
�ִ���ѧ�У�������ԭ�ӹ����ϵ���������������Ԫ�أ����ӵ��ܽ��ԣ�ͨ��������������ԭ�������ľ���ṹ������ʯī����������ԭ������������Ϊ5�����Է���sp3�ӻ������ڳɼ������ܹµ��ӶԵ��ų����ã����Բ�����ԭ�Ӳ���ͬһƽ���ϣ������б��ں��Ͱ��ף����ڲ���ԭ�Ӽ��γɹ��ۼ������Բ���Ӧ�γ�ԭ�Ӿ��塣�����廯���У�Bԭ�ӵ���������ȫ������ɼ������Է��ӳ�ƽ��ṹ���������У���ͼ�п��Կ�����ÿ��Bԭ���γ�4�����ۼ�����B�ļ۵���ֻ��3��������ÿ��Pԭ���ṩ1�Թ¶Ե�����Bԭ��(�ṩ�չ��)�γ���λ����������������ṹ����BP�����У�ÿ��Bԭ����2��Pԭ�ӹ��ɼн�Ϊ109��28���ĵ��������Σ��������Ҷ������ɽ��������߳���B-P������Ķ�����ϵ���Ӷ�����B-P������������ı߳������ս����ܶ���߳��ĵ�����ϵʽ��
(1)���ִ���ѧ�У�����ԭ�ӣ���ʹ��ԭ�ӹ��ף����ù����ϵ���������������Ԫ�ء�
�ڰ���(P4)�ĽṹΪ�������壬Ϊ�Ǽ��Է��ӣ������ڷǼ��Է���CS2�������ڼ��Է���ˮ��ԭ����P4��CS2�ǷǼ��Է��ӣ�H2O�Ǽ��Է��ӣ�������������ԭ����P4������ˮ��
��A������������ԭ������Χ3��Pԭ���γɹ��ۼ���ÿ��Pԭ�ӻ���1�Թ¶Ե��ӣ������ӻ���ʽΪsp3�ӻ���A��ȷ��
B���������в����֮�䲻�γɹ��ۼ�����ͨ�����»�����ϣ����Բ���������Ƿ��Ӽ���������B��ȷ��
C���������ÿһ������ԭ�ӷ���sp3�ӻ������ڹµ��ӶԵ��ų����ã�Pԭ�Ӳ���һ��ƽ���ϣ�C����ȷ��
D��PԪ�����ֳ����ĵ����У����ͺ����γɷ��Ӿ��壬�����ײ�����ԭ�Ӽ��γɹ��ۼ����൱��ԭ�Ӿ��壬���Ժ����۷е���ߣ�D��ȷ��
��ѡC��
��Ϊ��ԭ�ӹ��ף�P4��CS2�ǷǼ��Է��ӣ�H2O�Ǽ��Է��ӣ�������������ԭ����P4������ˮ��C��
(2)�����������У����þ�̯�����������Pԭ�Ӹ���Ϊ![]() =4����Bԭ�Ӹ���Ϊ4������ԭ�Ӹ�����Ϊ1:1����������Ļ�ѧʽΪBP���þ�����ԭ�Ӽ�ȫ���γɹ��ۼ���û�����������������Ծ���������ԭ�Ӿ��塣
=4����Bԭ�Ӹ���Ϊ4������ԭ�Ӹ�����Ϊ1:1����������Ļ�ѧʽΪBP���þ�����ԭ�Ӽ�ȫ���γɹ��ۼ���û�����������������Ծ���������ԭ�Ӿ��塣
�����廯������Ԫ�ػ�̬ԭ�Ӻ�������Ų�Ϊ2��8��18��7�������Ų�ʽΪ1s22s22p63s23p63d104s24p5��[Ar] 3d104s24p5�����廯������У�Bԭ�ӷ���sp2�ӻ��������ڹµ��Ӷԣ����Կռ乹����ƽ�������Σ����廯�����廯���ڸ������������з�Ӧ����BP��HBr���ϳ�����Ļ�ѧ����ʽΪ��PBr3��BBr3��3H2![]() BP��6HBr��
BP��6HBr��
����һ�������У���ԭ�ӵĵ��Ϸ�ʽΪ����һѭ����������ԭ�ӿռ�ѻ���ʽΪ�����������ܶѻ���ÿ����ԭ����Χ�����������ԭ����4����������ԭ�ӵ���λ��Ϊ4���ýṹ����һ����λ�����ṩ�չ����ԭ����B����֪������B��Pԭ�ӵ��������Ϊa nm������ȡһ����BΪ���ĵ��������壬Ȼ�������ȡһ�����������Σ���ʱ�����ļн�Ϊ109��28�����������Ҷ����ɵã�(![]() )2=a2+a2-2aacos109��28�������֪��cos109��28��= -
)2=a2+a2-2aacos109��28�������֪��cos109��28��= -![]() ���Ӷ����x=
���Ӷ����x=![]() nm����þ�����ܶȵı���ʽΪ
nm����þ�����ܶȵı���ʽΪ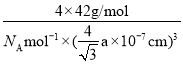 =
=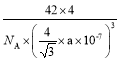 g/cm3��
g/cm3��
��Ϊ��BP��ԭ�Ӿ��壻1s22s22p63s23p63d104s24p5��[Ar] 3d104s24p5��ƽ�������Σ�PBr3��BBr3��3H2![]() BP��6HBr�������������ܶѻ���4��B��
BP��6HBr�������������ܶѻ���4��B��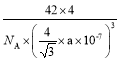 ��
��



| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�������ĵ���������BCl3��N2O��SiH4��SiHCl3�ȡ��ش��������⣺
(1)����B2H6��������Ͽ�������̬BCl3��ÿ����1.0 g BCl3�ų�5.9 kJ���������÷�Ӧ���Ȼ�ѧ����ʽΪ______��
(2)25��ʱ��Ӧ S(s) +H2(g)H2S(g) Kp1=6.0��105
Si(s)+2H2(g)SiH4(g) Kp2=7.8��106
��Ӧ Si(s)+2H2S(g)SiH4(g)+2S(s) Kp=_____(KpΪ�Է�ѹ��ʾ��ƽ�ⳣ������ͬ)��
(3)�ྦྷ�����з���һϵ�з�Ӧ��
(I)4SiHCl3(g)Si(s)+3SiCl4(g)+2H2(g) ��H1=a kJ /mol
(II)SiCl4(g)+H2(g)SiHCl3(g)+HCl(g) ��H2=b kJ/mo1
(III)SiCl2(g)+H2(g)Si(s)+2HCl(g) ��H3=c kJ/mo1
�ٷ�ӦSiHCl3 (g)SiCl2 (g) +HCl (g) ��H=____kJ/mo1 (�ú�a��b��c�Ĵ���ʽ��ʾ)
�ڷ�Ӧ(I)(II)(III)��Kp���¶ȵĹ�ϵ����ͼ
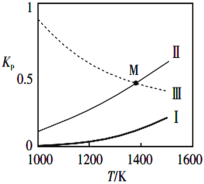
�������ȷ�Ӧ����_________(��I��II��III)��ͼ��M���ѹ�������ϵ��p(SiCl4)=______(��������ʵķ�ѹp��ʾ)��
(4)һ����ȡN2O�ķ���Ϊ O2NNH2(aq)��N2O(g)+H2O(1)���÷�Ӧ���������£�
(I)O2NNH2(aq)![]() O2NNH-(aq) +H+(aq) (����ƽ��)
O2NNH-(aq) +H+(aq) (����ƽ��)
(II)O2NNH-(aq)![]() N2O(g) +OH-(aq) (��)
N2O(g) +OH-(aq) (��)
(III)H+(aq)+OH-(aq)![]() H2O(1) (��)
H2O(1) (��)
�ٻ�����ķ�Ӧ������_________(��I��II��III)��
����֪��Ӧ(I)�����ʷ���v(��)=k1c(O2NNH2)��v(��)=k -1 c(O2NNH-)c(H+)��(k1��k -1�ֱ�Ϊ���淴Ӧ���ʳ�������Ӧ(I)�ﵽƽ��ʱ��ƽ�ⳣ��K=__________(��k1��k -1��ʾ)��
����֪�ܷ�Ӧ���ʷ���Ϊv=K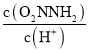 ����Ӧ(II)��v(��)=k2c(O2NNH-)����K=________ (��k1��k -1��k2��k3��ʾ)
����Ӧ(II)��v(��)=k2c(O2NNH-)����K=________ (��k1��k -1��k2��k3��ʾ)
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��һ����ͼ��ʾ����������ȼ�ϵ������������ˮ(��ˮ�и�Ũ���л�����C6H12O6��ʾ)��������������ȷ����
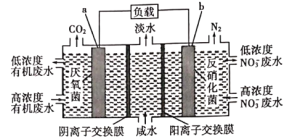
A.a�缫��ӦʽΪ��C6H12O6+6H2O-24e-��6CO2��+24H��
B.b�缫��������ҺpH����
C.�¶�Խ�ߣ�������ˮ��Ч��Խ��
D.����Ӧ��ת�Ƶĵ�����ΪNA�������ɱ�״����N2�����Ϊ2.24L
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����H2O2�ֽⷴӦ��Cu2+Ҳ��һ���Ĵ����á�Ϊ�Ƚ�Fe3+��Cu2+��H2O2�ֽ�Ĵ�Ч����ij��ѧ�о�С���ͬѧ�Ƿֱ��������ͼ�ס�����ʾ��ʵ��װ�á���ش�������⣺

(1)������ͼ��ͨ���۲�_________�����ԱȽϵó����ۡ���ͬѧ�����FeCl3��ΪFe2(SO4)3��Ϊ��������������_______��
(2)������ͼ����ʾװ�����������飬ʵ��ʱ��������40 mL����Ϊ����������Ӱ��ʵ������ؾ��Ѻ��ԡ�ʵ������Ҫ������������_______��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����(Se)����(Te)�ĵ��ʺͻ������ڻ��������ȷ��������ҪӦ�á�
(1)���й��������ڼ��仯�������������ȷ����_______��
A��Te λ�����ڱ��ĵ������� �� A ��
B��Se ��������ͨ����SeO2��SeO3
C��H2TeO4�����Ա�H2SO4������ǿ
D�����ȶ���H2Se��H2S��������HBrǿ
(2)25�� ʱ������ĵ���H2SeO4=H+ +![]() ��
��![]() H+ +
H+ +![]() Ka2 = 1��10-3����0.1 mol��L-1NaHSeO4��Һ��pHԼΪ________��NaHSeO4��Һ�е������غ����ʽΪ____________��
Ka2 = 1��10-3����0.1 mol��L-1NaHSeO4��Һ��pHԼΪ________��NaHSeO4��Һ�е������غ����ʽΪ____________��
(3)TeO2����ˮ�������ڽ�Ũ��ǿ���ǿ���ҵ�ϳ���ͭ������(��Ҫ��TeO2������������Ag��Au)Ϊԭ���Ʊ������ڣ��乤��������ͼ��
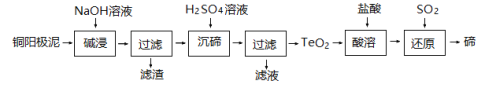
��ͭ�������ڼ��ǰ���ɡ��гɷ�ĩ��Ŀ����____________��
���������ʱTeO2������Ӧ�Ļ�ѧ����ʽΪ_____________��
����������ʱ������Һ�� pH Ϊ 4.5~5.0������TeO2���������Բ��ܹ�ǿ��ԭ����__________��
������ԭ���õ���̬��Ϊ���ڣ��Դ��ڽ���ϴ�ӣ��ж�ϴ�Ӹɾ���ʵ�������������________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����ж�һЩʵ����ʵ�����۽�����ȷ����
ѡ�� | ʵ����ʵ | ���۽��� |
A | HCl��������ˮ���ܵ��� | HClΪ���ӻ����� |
B | HBr������ǿ��HCl������ | Br�ķǽ����Ա�Clǿ |
C |
|
|
D | HF�ķе����HCl | F�ķǽ����Ա�Clǿ |
A.AB.BC.CD.D
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������Ԫ��X��Y��Z��Q��T��XԪ��Ϊ����Ԫ�أ�Xԭ�ӵ�M��p�ܼ���������δ�ɶԵ������չ����Yԭ�ӵļ۵����Ų�Ϊ3d64s2��Zԭ�ӵ�L���Ӳ��p�ܼ�����һ���չ����Qԭ�ӵ�L���Ӳ��P�ܼ���ֻ��һ�ԳɶԵ��ӣ�Tԭ�ӵ�M���Ӳ���p����������������������ȷ����
A.Ԫ��Y��Q���γɻ�����Y2Q3
B.X��T������������Ӧ��ˮ���ǰ�ߵ����ԱȺ���ǿ
C.X��Q������ɵĻ�����Ϊ���ӻ�����
D.ZQ2�Ǽ��Լ����ɵķǼ��Է���
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ͨ������£�NCl3��һ����״Һ�壬����ӿռ乹����NH3���ƣ����ж�NCl3��NH3���й�������ȷ����( )
A. ������N��Cl��������CCl4������C��Cl���������
B. NCl3�����ǷǼ��Է���
C. NBr3��NCl3�ӷ�
D. �ڰ�ˮ�У���NH3��H2O�����(����������ʾ)����γ�NH3��H2O���ӣ���NH3��H2O�ĽṹʽΪ![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����һ��������ܱ������У��������»�ѧ��Ӧ��CO2(g)��H2(g) ![]() CO(g)��H2O(g)���仯ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±���ʾ��
CO(g)��H2O(g)���仯ѧƽ�ⳣ��K���¶�t�Ĺ�ϵ���±���ʾ��
t �� | 700 | 800 | 830 | 1 000 | 1 200 |
K | 0.6 | 0.9 | 1.0 | 1.7 | 2.6 |
�ش��������⣺
��1���÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽΪK��_________________________________��
��2���÷�ӦΪ________��Ӧ(��������������������)��
��3��ij�¶��£������ʵ�ƽ��Ũ�ȷ�����ʽ��3c(CO2)��c(H2)��5c(CO)��c(H2O)�����жϴ�ʱ���¶�Ϊ______��
��4����830 ��ʱ���������г���1 mol CO��5 mol H2O����Ӧ�ﵽƽ����仯ѧƽ�ⳣ��K______1.0(������������С��������������)��
��5��830 ��ʱ�������еķ�Ӧ�Ѵﵽƽ�⡣�������������������£����������������ƽ��____�ƶ�(����������Ӧ�����������淴Ӧ��������������)��
��6����1 200 ��ʱ����ijʱ��ƽ����ϵ��CO2��H2��CO��H2O��Ũ�ȷֱ�Ϊ2 mol��L��1��2 mol��L��1��4 mol��L��1��4 mol��L��1�����ʱ������Ӧ��ƽ���ƶ�����Ϊ__________(��������Ӧ���������淴Ӧ�������������ƶ���)��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com