
����ѧ��--ѡ��3�����ʽṹ�����ʡ���15�֣�
����ͭ������̼��仯�����ڹ�ҵ�����Ź㷺��Ӧ�ã�
��1��Cu2������Χ�����Ų�ͼ�ɱ�ʾΪ ��
��2��Mn��̬ԭ�Ӻ����������ߵ��ܼ��ϵĵ��ӹ��� �ֲ�ͬ���˶�״̬��
��3����ͭ��������Ĵ��£�������CO��������CO2��HCHO��������CO2��H2O
�� N3-��CO2�ǵȵ����壬��N3-�ĽṹʽΪ ��
�� HCHO������Cԭ�ӹ�����ӻ�����Ϊ ��
��4����CuSO4��Һ�м������NaOH��Һ������[Cu(OH)4]2���������ǿռ乹�ͣ�[Cu(OH)4]2���Ľṹ����ʾ��ͼ��ʾΪ ��
��5�� �þ����x�������䷨���Բ�ð����ӵ��������Խ���ͭ�IJⶨ�õ����½��������Ϊ�����������ܶѻ����߳�Ϊ361pm(��ʾ��3.613=47.05������֪ͭ���ܶ�Ϊ9.00g��cm-3����ͭ������������
g��������λС�����������ӵ�����Ϊ (��ʽ���㣬������λС������
��1����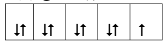 ��2�֣�
��2�֣�
��2��5��2�֣�
3d
��3����[N=N=N]-��2�֣� ��sp2 ��2�֣�
��4�� 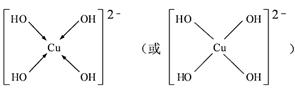 ��2�֣�
��2�֣�
��5�� 4.23��10-22g��2�֣� M(Cu)=64g/mol=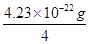 ��NA�� NA=6.05��1023mol-1��3�֣�
��NA�� NA=6.05��1023mol-1��3�֣�
���������������1��Cu2+3d����Ų���9�����ӣ�������Χ�����Ų�ͼ�ɱ�ʾΪ
��2��Mn��̬ԭ�Ӻ���������ߵ��ܼ�Ϊ3d���Ų���5�����ӣ���5�ֲ�ͬ���˶�״̬��
��3����N3-��CO2�ǵȵ����壬��N3-�Ľṹ��CO2���ƣ���ṹ��ʽΪ��[N=N=N]-��
��HCHO������Cԭ���γ���2��̼�ⵥ����1��̼��˫������3���Ҽ�������Cԭ���ӻ���ʽΪsp2
��4��OH?��Oԭ���ṩ�¶Ե��ӣ�Cu�ṩ�չ�����γ���λ��������[Cu(OH)4]2���Ľṹʾ��ͼΪ��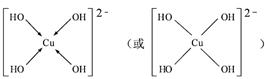
��5��Cu�ľ���Ϊ�����������ܶѻ�������Cuλ�ڶ���������ϣ�1��������Cu��8��1/8+6��1/2=4����ͭ�����������ǣ�4��64�£�6.02��1023��= 4.23��10-22g��һ������Ϊ�о�������M(Cu)=64g/mol=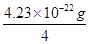 ��NA���ɵ� NA=6.05��1023mol-1��
��NA���ɵ� NA=6.05��1023mol-1��
���㣺���⿼������Ų����ȵ����塢�ӻ���ʽ����λ���������ļ��㡣


 ͬ����ϰǿ����չϵ�д�
ͬ����ϰǿ����չϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
�±�ΪԪ�����ڱ���һ����,�����Ԫ�آ١����ڱ��е�λ��,�û�ѧ����ش���������:
��1����֪�ɢ١��ۡ�������Ԫ����ɵ�ij�ֳ����������ˮ��Һ������,����ϸ������Ļ�����Ļ�ѧʽ����Ϊ��������������(����д����)��
��2���ɱ��Т١�������Ԫ�ذ�ԭ�Ӹ�����Ϊ1��1���ɵĻ������ϡ��Һ�ױ����ֽ�,ͨ��ʹ�õĴ���Ϊ������ (�����)��
a�� MnO2 b�� FeCl3 c��Na2SO3 d��KMnO4
��3����ͼ��A��F���ɲ����ϱ���Ԫ����ɵĵ��ʻ���,����A��B��CΪ����,ת����ϵ����:
����BΪ��ɫ����,AΪԭ�Ӱ뾶��С��ԭ����ɵĵ��ʡ�
��F�ĵ���ʽΪ����������
��ʵ������ʼ�μӷ�Ӧ��B��������ɵ�B�������,����ʼ�μӷ�Ӧ��A��C�����ʵ���֮���� �� ��
����DΪ����ɫ����,��ɫ��ӦΪ��ɫ,���C��Ԫ�ص�ԭ���������������ڲ��������2����
�����й���D��˵����ȷ������������ (����ĸ)��
a������ˮ�������Ϸ�Ӧ b������������,���л�ԭ��
c���Ⱥ����Ӽ�,�ֺ��Ǽ��Թ��ۼ� d����һ�ּ���������
���ö��Ե缫��F�ı�����Һ���е��,��������Ӧʽ��������������������������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��������(NaN3)��һ����ɫ���壬�����������Ʊ�����Ϊ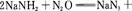
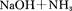
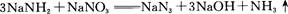 �ش��������⣺
�ش��������⣺
��1�������ڵ������У��縺������Ԫ���� ����һ��������С��Ԫ���� ��
��2����̬��ԭ�ӵ�L������Ų�ͼΪ ��
��3����N3-��Ϊ�ȵ�����ķ���Ϊ (д��һ��)�����ݼ۲���ӶԻ������ۣ�NO3-�Ŀռ乹��Ϊ ��
��4����������(NaNH2)�͵�������(NaN3)�ľ�������Ϊ ���������Ƶ�ˮ��Һ�ʼ��ԣ������ӷ���ʽ��ʾ��ԭ�� ��
��5��N20�е�(һ88.49���NH3�е�(һ33.34��)�ͣ�����Ҫԭ����
��6����ȫ���ҵ����ԭ��Ϊ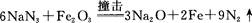
�ٵ������Цļ��ͦм���Ŀ֮��Ϊ ��
���������д��ڵĻ�ѧ������Ϊ ��
��������Ϊ���������ѻ����侧����ͼ��ʾ�������߳�Ϊa cm�����������ܶ�Ϊ
(�ú�a��NA�ı���ʽ��ʾ������NAΪ�����ӵ�����)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��8�֣����������л�Ϊͬ���칹����� ����Ϊͬ����������� ������ͬλ�ص��� ������ͬһ�����ʵ��� ��(�����)
��Һ�� �� �۰��� ��18O ��
�۰��� ��18O ��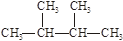
���� ��16O ������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ-ѡ��3���ʽṹ�����ʡ���15�֣�
��ش��������⣺
��1���������ڵ�ij����Ԫ�أ����һ�����������������ͼ1��ʾ�����Ԫ�ض�Ӧԭ�ӵ�M������Ų�ʽΪ ��
��2������ͼ2��ʾ��ÿ�����߱�ʾ���ڱ���A-��A�е�ijһ��Ԫ���⻯��ķе�仯��ÿ��С�ڵ����һ���⻯�����a��������� ����������ж����� ��
��3��CO2�ڸ��¸�ѹ�����γɵľ����侧������ͼ3��ʾ���þ������������ ��ѡ����ӡ���ԭ�ӡ������ӡ������������壬�þ�����̼ԭ�ӹ�����ӻ�����Ϊ ��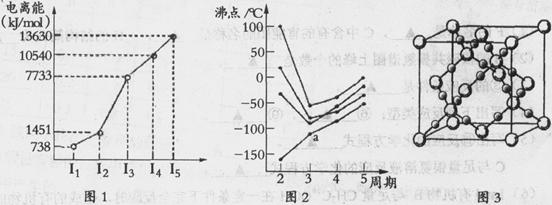
��4�������Ӿ��������������Ӽ������ܶ�ĽӴ����Խ�����ϵ��������ʹ�����ȶ����ڡ���֪Na+�뾶��Cl-��a����Cs+�뾶��Cl-��b������ع˿α���NaCl��CsCl�ľ������侧���߳���Ϊ ��
��5��Fe��һ�־�����ס�����ʾ�����������߷������ҵõ���A-Dͼ����ȷ���� ����ԭ�ӵ���λ���� ��������ԭ�ӵİ뾶��r cm���þ�����ܶ���p g��cm3�����������ԭ������Ϊ ���谢���ӵ�������ֵΪNA����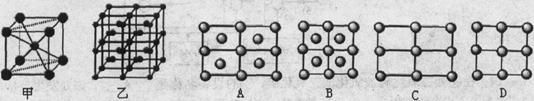 ��
��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
����ѧ����ѡ��3�����ʽṹ�����ʡ���15�֣�
��֪A��B��C��D��E��Ԫ�����ڱ���1��36��Ԫ�أ���ԭ����������������A��B��C��DΪ����Ԫ�ء�A����������Ԫ�ؼȲ���ͬһ���ڣ�Ҳ����ͬһ���塣B��Cͬ���壬D��Eͬ���ڣ�Ԫ��E�����ڱ��еĵ�7��Ԫ�أ�D�����������������ڲ��������ͬ��D��B���γ����ӻ�����侧���ṹ(����Bԭ���ھ����ڲ�)��ͼ��
��ش��������⣺
��1��C-�ĵ����Ų�ʽΪ______________��EԪ��ԭ�ӵļ۵����Ų�ʽΪ________��
��2��Ԫ��B�뵪Ԫ�ؿ����γɻ�����NB3������N��B���Ļ�ѧ������Ϊ________�����ݼ۲���ӶԻ������ۿ����ж�NB3�Ŀռ乹��Ϊ________��NB3������Nԭ�ӵ��ӻ���ʽΪ________�ӻ���
��3��A��B�γɵĻ�����AB����Է���������A��C�γɵĻ�����AC����Է�������С����AB�ķе��AC�ߣ���ԭ���� ��
��4���Ӿ���ͼ���Եó���D��B�γɵ����ӻ�����Ļ�ѧʽΪ ��
��5������DԪ�صĻ�������ɫ��ӦΪ______ɫ����ɫ��Ӧ��ԭ���� ��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��15�֣���֪A��B��C��D���ֶ�����Ԫ�أ����ǵĺ˵������������Aԭ�ӡ�Cԭ�ӵ�L�ܲ��У���������δ�ɶԵĵ��ӣ�C��Dͬ���塣 E��F���ǵ�������Ԫ�أ�Eԭ�Ӻ�����4��δ�ɶԵ��ӣ�Fԭ�ӳ������ܲ�ֻ��1�������⣬������ܲ��Ϊȫ����������������Ϣ��գ�
�Ż�̬Dԭ���У�����ռ�ݵ�����ܲ���� �����ܲ���е�ԭ�ӹ����Ϊ ��
��E2�����ӵļ۲�����Ų�ͼ�� ��Fԭ�ӵĵ����Ų�ʽ�� ��
��AԪ�ص�����������Ӧ��ˮ��������ԭ�Ӳ�ȡ�Ĺ���ӻ���ʽΪ �� BԪ�ص���̬�⻯���VSEPRģ��Ϊ ��
�Ȼ�����AC2��B2C��������DAB����Ϊ�ȵ����壬���ǽṹ���ƣ�DAB���ĵ���ʽΪ ��
����������ɫ��Ӧ����ɫ�����ڽ�����������E3������λ��AB�����ɣ���λ��Ϊ6����ˮ��Һ��������ʵ������E2�����ӵĶ��Լ��飬����E2�����ӵ����ӷ���ʽΪ ��
��ij�ֻ�������D��E��F����Ԫ����ɣ��侧����ͼ��ʾ�����仯ѧʽΪ ���þ������µ���Ϊ�����Σ���������洹ֱ������ͼ����ʾ��������ʽ����þ�����ܶȣ�
d= g/cm3��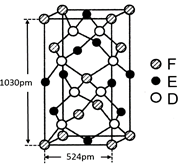
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
(12�֣�ÿ��2�֣�������(Na3N)�ǿ�ѧ���Ʊ���һ����Ҫ���������ˮ���ÿɲ���NaOH��NH3����ش��������⣺
��1��Na3N�ĵ���ʽ��_______���û���������______���γɵ�_____������ӡ����ۡ��������
��2��Na3N�����ᷴӦ����__________���Σ���Ӧ�ķ�Ӧ����ʽ�� ��
��3���Ƚ�Na3N���������İ뾶��r(Na+)__________r(N3��)(�������=������������)��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
��1��д����ԭ�ӵ�ԭ�ӽṹʾ��ͼ
��2����Ԫ�ص�ԭ���ڷ�Ӧ���� ����á���ʧ���� �����ӣ����ֳ� �ԡ�
��3����Ԫ�ص�����ϼ�Ϊ ����ͻ��ϼ�Ϊ ��
��4����Ԫ�صķǽ����� ���ǿ��������������Ԫ�صķǽ����ԡ�
��5��д����Ԫ�ء���Ԫ������������Ӧˮ����Ļ�ѧʽ���Ƚ�����ǿ�� ��
��6����д���������ʵĵ���ʽ��NaCl H2O CO2 N2 ��
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com