
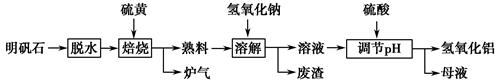
 Al2(SO4)3£«
Al2(SO4)3£« S
S
 Al2O3£«
Al2O3£« ________”ü”£
________ӟӣ


| Äź¼¶ | øßÖŠæĪ³Ģ | Äź¼¶ | ³õÖŠæĪ³Ģ |
| øßŅ» | øßŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” | ³õŅ» | ³õŅ»Ćā·ŃæĪ³ĢĶĘ¼ö£” |
| ø߶ž | ø߶žĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õ¶ž | ³õ¶žĆā·ŃæĪ³ĢĶĘ¼ö£” |
| øßČż | øßČżĆā·ŃæĪ³ĢĶĘ¼ö£” | ³õČż | ³õČżĆā·ŃæĪ³ĢĶĘ¼ö£” |
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā

| ŹµŃé²Ł×÷ | Ź±¼ä/min | µē×ÓĢģĘ½µÄ¶ĮŹż/g |
| ÉÕ±£«NaOHČÜŅŗ | 0 | 120 |
| ÉÕ±£«NaOHČÜŅŗ£«ŃłĘ· | 0 | 135 |
| 1 | 134.5 | |
| 2 | 134.1 | |
| 3 | 133.8 | |
| 4 | 133.8 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗŹµŃéĢā
 ²ā¶ØŹ£Óą¹ĢĢåÖŹĮ棬ŹµŃéÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß”£
²ā¶ØŹ£Óą¹ĢĢåÖŹĮ棬ŹµŃéÖŠ·¢Éś·“Ó¦µÄ»Æѧ·½³ĢŹ½ŹĒ£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß£ß”£ ²ā¶ØÉś³ÉĘųĢåµÄĢå»żŹµŃé×°ÖĆ£ŗ
²ā¶ØÉś³ÉĘųĢåµÄĢå»żŹµŃé×°ÖĆ£ŗ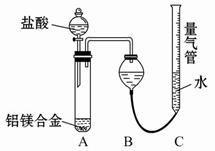

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗĢīæÕĢā
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®AlŹĒµŲæĒÖŠŗ¬Įæ×ī¶ąµÄŌŖĖŲ |
| B£®AlŹĒ±Č½Ļ»īĘĆµÄ½šŹō£¬ŌŚ»Æѧ·“Ó¦ÖŠČŻŅ׏§Č„µē×Ó£¬±ķĻÖŃõ»ÆŠŌ |
| C£®ÓėAl·“Ó¦²śÉśH2µÄČÜŅŗŅ»¶Ø³ŹĖįŠŌ |
| D£®Al²ŌŚæÕĘųÖŠŹÜČČæÉŅŌČŪ»Æ£¬ÓÉÓŚŃõ»ÆĤµÄ“ęŌŚ£¬ČŪ»ÆµÄAl²¢²»µĪĀä |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā

²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
| A£®¢ŁÖŠ²śÉśĘųÅŻ£¬¢ŚÖŠ²»²śÉśĘųÅŻ |
| B£®¢ŁŗĶ¢ŚÖŠ¶¼²»²śÉśĘųÅŻ |
| C£®¢ŁŗĶ¢ŚÖŠ¶¼²śÉśĘųÅŻ£¬¢ŁÖŠŹĒH2£¬¢ŚÖŠŹĒO2 |
| D£®¢ŁŗĶ¢ŚÖŠ¶¼²śÉśĘųÅŻ£¬ĒŅ¶¼ŹĒH2 |
²éæ““š°øŗĶ½āĪö>>
æĘÄæ£ŗøßÖŠ»Æѧ Ą“Ō“£ŗ²»Ļź ĢāŠĶ£ŗµ„Ń”Ģā
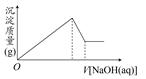
| A£®ÓŠMg2£«£¬Ć»ÓŠAl3£« |
| B£®ÓŠAl3£«£¬Ć»ÓŠMg2£« |
| C£®ÓŠ“óĮæµÄH£«”¢Mg2£«ŗĶAl3£« |
| D£®ÓŠMg2£«ŗĶAl3£« |
²éæ““š°øŗĶ½āĪö>>
°Ł¶ČÖĀŠÅ - Į·Ļ°²įĮŠ±ķ - ŹŌĢāĮŠ±ķ
ŗž±±Ź”»„ĮŖĶųĪ„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±ØĘ½ĢØ | ĶųÉĻÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | µēŠÅÕ©Ę¾Ł±Ø×ØĒų | É꥜Ź·ŠéĪŽÖ÷ŅåÓŠŗ¦ŠÅĻ¢¾Ł±Ø×ØĒų | ÉęĘóĒÖČؾŁ±Ø×ØĒų
Ī„·ØŗĶ²»Į¼ŠÅĻ¢¾Ł±Øµē»°£ŗ027-86699610 ¾Ł±ØÓŹĻä£ŗ58377363@163.com