
| I1/kJ•mol-1 | I2/kJ•mol-1 | I3/kJ•mol-1 | I4/kJ•mol-1 | I5/kJ•mol-1 |
| 738 | 1451 | 7733 | 10540 | 13630 |
���� ��1����Clԭ�Ӻ��������Ϊ17����̬ԭ�Ӻ�������Ų�Ϊ1s22s22p63s23p5���ݴ˽��
�ڸ��ݼ۲���ӶԻ�������ȷ�����ӿռ乹�͡�Bԭ�ӵ��ӻ�������ͣ�
��Ԫ�صķǽ�����Խǿ����縺��Խ��
��2���ٺ�������Ų���ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС��
�ڸ�Ԫ�صĵ�III�����ܾ��������Ԫ�����ڵ�IIA�壻
��3��NaH����NaCl�;���ṹ��ʳ�ξ�����Na+��Cl-�ļ��Ϊ�ⳤ��һ�룬�ݴ˷������
��� �⣺��1���ٻ�̬Clԭ���е���ռ�ݵ�����ܲ�Ϊ��3�ܲ㣬����M�����ܲ���1��s�����3��p�����5��d���������9 ��ԭ�ӹ����
�ʴ�Ϊ��M��9��
��BH4-��Bԭ�Ӽ۲������=4+$\frac{1}{2}$����3+1-4��1��=4��Bԭ�ӵ��ӻ����������sp3�ӻ����Ҳ����µ��Ӷԣ��������������幹�ͣ�
�ʴ�Ϊ���������壻sp3��
�۷ǽ����ķǽ�����Խǿ��縺��Խ�ǽ�������ǿ����HԪ�أ������BԪ�أ���С����LiԪ�أ�����Li��B��HԪ�صĵ縺���ɴ�С����˳��ΪH��B��Li��
�ʴ�Ϊ��H��B��Li��
��2���ٺ�������Ų���ͬ���˵����Խ�����Ӱ뾶ԽС��﮵�������Ϊ3�����������Ϊ1��Li+��H-�����������Ϊ2�����뾶Li+��H-��
�ʴ�Ϊ������
�ڸ�Ԫ�صĵ�III�����ܾ��������Ԫ�����ڵ�IIA�壬ΪMgԪ�أ�
�ʴ�Ϊ��Mg��
��3��NaH����NaCl�;���ṹ��NaH����ľ�������a=488pm���ⳤ����Na+�뾶Ϊ102pm��H-�İ뾶Ϊ$\frac{488-102��2}{2}$=142pm���þ����������Ӹ���=8��$\frac{1}{8}$+6��$\frac{1}{2}$=4�������Ӹ���=12��$\frac{1}{4}$+1=4��NaH�������ܶ��Ǧ�=$\frac{4M}{{N}_{A}V}$=$\frac{24��4}{6.02��1{0}^{23}��48{8}^{3}��1{0}^{-30}}$��1.37��
�ʴ�Ϊ��142pm��1.37��
���� ���⿼�����ʽṹ�����ʣ��漰��������Ų����ӻ���ʽ���жϡ��ռ乹�͵��жϡ������ļ����֪ʶ�㣬�ѵ��Ǿ����ļ��㣬������ù�ʽ�ǽⱾ��ؼ����Ѷ��еȣ�


 ��У���˳�̾�ϵ�д�
��У���˳�̾�ϵ�д� ��У���һ��ͨϵ�д�
��У���һ��ͨϵ�д� �γ̴����Ծ�����100��ϵ�д�
�γ̴����Ծ�����100��ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
| ���� | K2CO3 | KHCO3 | K2SO4 | KMnO4 |
| �ܽ��g/100g | 111 | 33.7 | 11.1 | 6.34 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���ۺ���ά�صĻ�ѧʽ��Ϊ��C6H10O5��n���ʻ�Ϊͬ���칹�� | |
| B�� | ú��������Һ�������ú�������ʡ�������Ⱦ | |
| C�� | ����Al2O3�����������������������ռ� | |
| D�� | ú�ĸ���ʯ�͵ķ���Ϊ�����仯 |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | ���������Ƿ�ˮ�⣺������Һ��ϡ���������ˮԡ����һ��ʱ�������������Һ��ϼ��ȣ��۲����� | |
| B�� | ������Һ���Ƿ���NH4+��ȡ������������Һ�������м���ŨNaOH��Һ���ȣ�����ʪ��ĺ�ɫʯ����ֽ�����Թܿڣ��۲����� | |
| C�� | �ᴿ�����������ӵı��������������ӵı��������Ũ��ˮ�����ù��ˣ���ȥ���屽�ӳ��� | |
| D�� | ̽����ѧ��Ӧ���ȣ�ȡ5mL0.1mol/LKI��Һ���μ�0.1mol/LFeCl3��Һ5��6�Σ���ַ�Ӧ���ɸ�����Һ�м���I2�ֺ�I-��ʵ����ʵ�жϸ÷�Ӧ�ǿ��淴Ӧ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ������
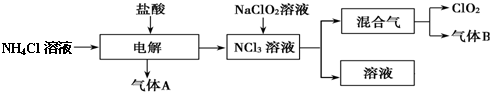
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | Cu��Cu��ϡ���� | B�� | C��Pt���Ȼ�����Һ | C�� | Cu��Zn���ƾ� | D�� | Fe��Cu��FeCl3��Һ |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| A�� | C4H10��3��ͬ���칹�� | |
| B�� | ������������黥Ϊͬ���칹�壬���Ƿ�����������ѧ���������������ȫ��ͬ | |
| C�� | ����Ķ����������������������� | |
| D�� | ����ʽΪC6H6������һ���DZ� |
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�ѡ����
| ѡ�� | ���� | ���� | ���� |
| A | �� | ���� | ��Ũ��ˮ����� |
| B | �� | �ױ� | ���μ������Ը��������Һ��NaOH��Һ���Һ |
| C | �屽 | Br2 | ��NaOH��Һ���Һ |
| D | �������� | ���� | �ӱ���̼������Һ���Һ |
| A�� | A | B�� | B | C�� | C | D�� | D |
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com