
����Ŀ���й����ʵ�ת����ϵ����ͼ��ʾ���������ʺ���������ȥ����A��������Ԫ����ɵ�������ˮ�����ʣ�����Ԫ�ص�������Ϊ7:4��B��ǿ�C��Ħ������Ϊ34g��mol-1�� F���������ɫҺ�壻��ɫ����G��ʹƷ����Һ��ɫ������I����Һ�еμ�KSCN��Һ����Һ����Ѫ��ɫ��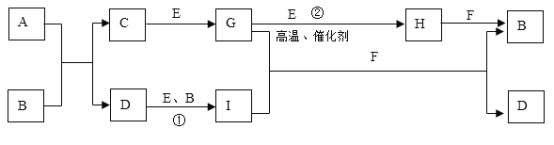
��ش��������⣺
��1��A�Ļ�ѧʽ____________________________��
��2��C�ĵ���ʽ____________________________��
��3����Ӧ�ٵ����ӷ���ʽ_______________________________________________________��
��4����Ӧ�ڵĻ�ѧ����ʽ_______________________________________________________��
���𰸡� FeS ![]() 4Fe2+ + O2 +4H+ ==4Fe3+ + 2H2O 2SO2 + O2
4Fe2+ + O2 +4H+ ==4Fe3+ + 2H2O 2SO2 + O2 ![]() 2SO3
2SO3
��������GΪ�����ҿ�ʹƷ����ɫ��GΪSO2������C��Ħ������Ϊ34g��mol-1��֪CΪH2S��EΪO2��HΪSO3��FΪH2O��BΪH2SO4������I����Һ�еμ�KSCN��Һ����Һ����Ѫ��ɫ��IΪFe2(SO4)3���Ӷ�DΪFeSO4������Ԫ���غ��֪A�к���S��FeԪ�ء�
��1��A�к���Fe��S����Ԫ�أ�ԭ�Ӹ�����Ϊ(7/56):(4/32)=1:1������A�Ļ�ѧʽΪFeS��
��2��CΪH2S������ʽΪ![]() ��
��
��3��D��E��B��I�ֱ�ΪFeSO4��O2��H2SO4��Fe2(SO4)3��Ӧ�����ӷ���ʽΪ��4Fe2+ + O2 +4H+ ==4Fe3+ + 2H2O��
��4����Ӧ����SO2��O2������������Ӧ����SO3����ѧ����ʽΪ��2SO2 + O2 ![]() 2SO3��
2SO3��


 ��ѧ����ϵ�д�
��ѧ����ϵ�д� �ο�������ϵ�д�
�ο�������ϵ�д�
| �꼶 | ���пγ� | �꼶 | ���пγ� |
| ��һ | ��һ��ѿγ��Ƽ��� | ��һ | ��һ��ѿγ��Ƽ��� |
| �߶� | �߶���ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
| ���� | ������ѿγ��Ƽ��� | ���� | ������ѿγ��Ƽ��� |
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��������X����Է�������Ϊ92����XΪ��ʼԭ�Ϻϳ�E��·������(���ֲ���ͷ�Ӧ��������ȥ)��

�ش��������⣺
��1���л���X�Ľṹ��ʽΪ_________________��
��2��A��B�ķ�Ӧ����Ϊ________________________________��
��3��C��Dת���ТٵĻ�ѧ����ʽΪ________________________________��
��4�����й���B��˵����ȷ����____________��
a���˴Ź���������5���� b���ܷ�����ȥ��Ӧ�ͻ�ԭ��Ӧ
c�������Ʒ�Ӧ�ų����� d���ܺ�NaOH��Һ��Ӧ
��5��E�ж���ͬ���칹�壬ͬʱ��������������ͬ���칹�干��_______�֡�
����NaHCO3��Һ������ų� �ڷ����廯����
���в��������л���Ľṹ��ʽΪ_________��
��6���л���X��ͨ�������ϳ�W��W�Ǹ߷��ӻ�����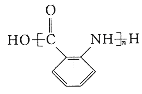 �ĵ��塣
�ĵ��塣
��֪�������ϵļ����Ա�����KMnO4�������Ȼ�
���������������ɱ�Fe/HCl��ԭΪ���������������İ����ױ�����

��F�Ľṹ��ʽΪ_______________���Լ�aΪ_______________��
����һ��������W�ۺϳɸ߷��ӻ�����Ļ�ѧ����ʽΪ___________________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��ij����ѧϰС����ѧϰ��![]() ��
��![]() �ķ�Ӧ����Ϊ
�ķ�Ӧ����Ϊ![]() ��
��![]() Ӧ��Ҳ���Է�Ӧ�������������ͼװ��
Ӧ��Ҳ���Է�Ӧ�������������ͼװ��![]() �г�װ������ȥ��װ�õ�����������
�г�װ������ȥ��װ�õ�����������![]() ����ʵ�飬̽��
����ʵ�飬̽��![]() ��
��![]() ��Ӧ�IJ���밴Ҫ��ش��������⡣
��Ӧ�IJ���밴Ҫ��ش��������⡣
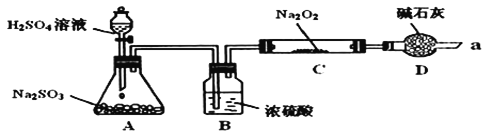
��![]() д��װ��A�з�����Ӧ�Ļ�ѧ����ʽ��______��
д��װ��A�з�����Ӧ�Ļ�ѧ����ʽ��______��
��![]() װ��D�����ã����˿��Է�ֹ�����е�
װ��D�����ã����˿��Է�ֹ�����е�![]() ��ˮ�����Ƚ���C����
��ˮ�����Ƚ���C����![]() ��Ӧ��������______________________________________________________��
��Ӧ��������______________________________________________________��
��![]() ��ͨ��������
��ͨ��������![]() ��
��![]() ��ַ�Ӧ�����Ƕ�C�й������������¼��裺
��ַ�Ӧ�����Ƕ�C�й������������¼��裺
����1��ֻ��![]() ��
��
����2��___________��
����3������![]() ������
������![]() ��
��
��1��������2�������![]() ��
��![]() ��Ӧ�Ļ�ѧ����ʽ��_________________��
��Ӧ�Ļ�ѧ����ʽ��_________________��
��2��ijͬѧ���������ʵ���һ��ȷ�ϲ���ijɷ֡�
ʵ�鲽�� | ���� |
| ����ȫ���ܽ� |
| ���� |
| ������ɫ���� |
����![]() �н�����������ͨ����������
�н�����������ͨ����������![]() ��Һ�У�������Ӧ�����ӷ���ʽΪ��______��ͨ������ʵ������ȷ������______������
��Һ�У�������Ӧ�����ӷ���ʽΪ��______��ͨ������ʵ������ȷ������______������![]() ѡ��1��2��
ѡ��1��2��![]()
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����C��H��O��ɵĻ�����8.80mg����ȫȼ�պ�ö�����̼22.0mg��ˮ10.8mg���û������ʵ��ʽ�����ʽ���ǣ� ��
A. C5H6OB. C5H12C. C5H12O2D. C5H12O
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ�����о�����ԭ��ֱ�ӹ���,�����ڷ��Ӿ������( )
A.��̬�� B.��̬��
C.�� D.��������
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ��SO2�dz�������������ڻ����������������Ӧ�ù㷺��
I��ͼ��ʵ������ȡSO2����֤SO2ijЩ���ʵ�װ��ͼ
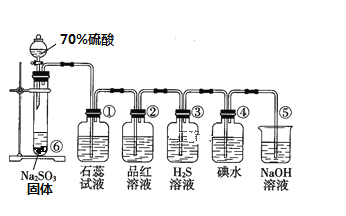
��1�����з�����Ӧ�Ļ�ѧ����ʽΪ_________________________________
��2������ʵ������Ϊ_____________________________________��֤��SO2��____��
��3���������ӷ�Ӧ����ʽΪ______________________
IIijѧ����SO2��Ư�۾��ķ�Ӧ����ʵ��̽����
���� | ���� |
ȡ4gƯ�۾����壬����100mLˮ | ���ֹ����ܽ⣬��Һ������ɫ |
���ˣ���Ư�۾���pH | pH��ֽ�ȱ�����ԼΪ12��������ɫ |
| i.Һ���Ϸ����ְ��� ii.�Ժ��ֻ��ǣ���Һ��Ϊ����ɫ iii.�Ժ���������ɫ����������ɫ��ȥ |
��1��pH��ֽ��ɫ�ı仯˵��Ư�۾���Һ���е�������_________________
��2����ˮ�г���ͨ��SO2��δ�۲쵽�������Ʋ�����i�İ�����HClСҺ���γɣ���������ʵ�飺
a����ʪ��ĵ⻯�ص�����ֽ����������ޱ仯��
b�����ữ��AgNO3��Һ���������������ɫ������
��ʵ��a��Ŀ����_______________________________________________
����ʵ��a��b�����жϰ����к���HCl��������_______________________________________
��3����Aƿ�л������ˡ�ϴ�ӣ��õ�����X
�������X�м���ϡ���ᣬ�����Ա仯��ȡ�ϲ���Һ������BaCl2��Һ��������ɫ�����������X�к��е�������___________
�������ӷ���ʽ��������iii�л���ɫ��ȥ��ԭ�� ___________________________
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ���ƺ��ƵĻ�������������Ҫ����;��
��1��̼���ƿ����ڴӺ�ˮ��ȡ�壬�漰�ķ�Ӧ���£�3Br2+6Na2CO3+3H2O=5NaBr+NaBrO3+6NaHCO3,��������Ӧ����˫���ŷ��������ת�Ƶķ������Ŀ_______________���÷�Ӧ���������뻹ԭ�������ʵ���֮���� __________��
��2����������Ӧ������_________�У����Ż�ʱӦ��ȡ������ʩ��_______��
��3������������DZͧ������ʱ�����Ļ�ѧ��Ӧ����ʽ��__________________���÷�Ӧ������������_____________��
��4�������д�ŵĴ����С�մ������������ʱ����������ó������е��������������ǵķ���_________________��
�鿴�𰸺ͽ���>>
��Ŀ�����л�ѧ ��Դ�� ���ͣ�
����Ŀ����1����һ���ƿ����Ϊm1����ƿ�������������Ϊm2������ͬ״���£�����Ϊ����ij����Aʱ��������Ϊm3����֪������Է�������Ϊ29����A����Է���������_______________
��2����һ�������۷����Ȼ�����Һ�У���ȫ��Ӧ�õ���Һ��Fe2+��Fe3+��Ũ��ǡ����ȡ����ѷ�Ӧ��Fe3+��δ��Ӧ��Fe3+�����ʵ���֮��Ϊ___________
��3����֪ͭ��ϡ���ᷴӦ����ʽΪ3Cu+8HNO3=3Cu��NO3)2+2NO��+4H2O���������������뱻��ԭ������������Ϊ___________
��4��A��B������ƿ�зֱ�װ80mL 3.0 mol/L�������NaOH��Һ���ٷֱ�ӵ����������ۣ�����ͬ״���²���������������ΪA:B=2:5����������۵�����Ϊ___________
�鿴�𰸺ͽ���>>
����ʡ������Υ���Ͳ�����Ϣ�ٱ�ƽ̨ | �����к���Ϣ�ٱ�ר�� | ����թƭ�ٱ�ר�� | ����ʷ���������к���Ϣ�ٱ�ר�� | ������Ȩ�ٱ�ר��
Υ���Ͳ�����Ϣ�ٱ��绰��027-86699610 �ٱ����䣺58377363@163.com