
题目列表(包括答案和解析)
C(s)+O2(g)![]() CO2(g);ΔH=-393.51 kJ·mol-1②
CO2(g);ΔH=-393.51 kJ·mol-1②
计算下列反应的反应热ΔH:C(s)+CO2(g) ![]() 2CO(g)( )?
2CO(g)( )?
A.-283.01 kJ·mol? B.172.51 kJ·mol-1??
C.283.1 kJ·mol-1?? D.504.00 kJ·mol-1??
已知:①C(s) +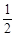 O2(g)
O2(g) CO(g);△H =" –" 110.5 kJ · mol – 1
②C(s) + O2(g) CO2(g);△H =" –" 393.51 kJ · mol – 1
则反应:C(s) + CO2(g) 2CO(g)的△H为 ( )
| A.– 283.01 kJ · mol – 1 | B.+ 172.51 kJ · mol – 1 |
| C.+ 283.01 kJ · mol – 1 | D.+ 504.00 kJ · mol – 1 |
已知:①C(s) + O2(g)
O2(g) CO(g);△H =" –" 110.5 kJ · mol – 1
②C(s) + O2(g)
CO2(g);△H ="
–" 393.51 kJ · mol – 1
则反应:C(s) + CO2(g) 2CO(g)的△H为 ( )
A.– 283.01 kJ · mol – 1 B.+ 172.51 kJ · mol – 1
C.+ 283.01 kJ · mol – 1 D.+ 504.00 kJ · mol – 1
已知:①C(s) + O2(g)
O2(g)
CO(g);△H = – 110.5 kJ · mol – 1
②C(s) + O2(g) CO2(g);△H =
– 393.51 kJ · mol –
1
则反应:C(s) + CO2(g) 2CO(g)的△H为 ( )
A.– 283.01 kJ · mol – 1 B.+ 172.51 kJ · mol – 1
C.+ 283.01 kJ · mol – 1 D.+ 504.00 kJ · mol – 1
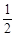 O2(g)
O2(g) | A.– 283.01 kJ · mol – 1 | B.+ 172.51 kJ · mol – 1 |
| C.+ 283.01 kJ · mol – 1 | D.+ 504.00 kJ · mol – 1 |
湖北省互联网违法和不良信息举报平台 | 网上有害信息举报专区 | 电信诈骗举报专区 | 涉历史虚无主义有害信息举报专区 | 涉企侵权举报专区
违法和不良信息举报电话:027-86699610 举报邮箱:58377363@163.com