
题目列表(包括答案和解析)
25℃、101kPa下,碳、氢气和甲烷的燃烧热依次是393.5 kJ·mol—1、285.8 kJ·mol—1
和890.3kJ·mol—1,下列选项中正确的是
| A.2H2(g)+O2(g)=2H2O(l)△H=—285.8kJ·mol—1 |
| B.CH4(g)+2O2(g)=CO2(g)+2H2O(l)△H=+890.3kJ·mol—1 |
| C.H2(g)+1/2O2(g)=H2O(g)△H<—285.8kJ·mol—1 |
| D.C(s)+2H2(g)=CH4(g)△H=—74.8 kJ·mol—1 |
| A.2H2(g)+O2(g)=2H2O(l)△H=—285.8kJ·mol—1 |
| B.CH4(g)+2O2(g)=CO2(g)+2H2O(l)△H=+890.3kJ·mol—1 |
| C.H2(g)+1/2O2(g)=H2O(g)△H<—285.8kJ·mol—1 |
| D.C(s)+2H2(g)=CH4(g)△H=—74.8 kJ·mol—1 |
25℃、101kPa下,碳、氢气、甲烷和葡萄糖的燃烧热依次是ΔH=-393.5kJ/mol、ΔH=-285.8kJ/mol、ΔH=-890.3kJ/mol、ΔH=-2800kJ/mol,则下列热化学方程式正确的是( )
A.C(s)+ O2(g)===CO(g)ΔH=-393.5kJ/mol O2(g)===CO(g)ΔH=-393.5kJ/mol |
| B.2H2(g)+O2(g)===2H2O(g)ΔH=+571.6kJ/mol |
| C.CH4(g)+2O2(g)===CO2(g)+2H2O(g)ΔH=-890.3kJ/mol |
D. C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l)ΔH=-1400kJ/mol C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l)ΔH=-1400kJ/mol |
25℃、101kPa下,碳、氢气、甲烷和葡萄糖的燃烧热依次是ΔH=-393.5kJ/mol、ΔH=-285.8kJ/mol、ΔH=-890.3kJ/mol、ΔH=-2800kJ/mol,则下列热化学方程式正确的是( )
A.C(s)+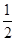 O2(g)===CO(g)ΔH=-393.5kJ/mol
O2(g)===CO(g)ΔH=-393.5kJ/mol
B.2H2(g)+O2(g)===2H2O(g)ΔH=+571.6kJ/mol
C.CH4(g)+2O2(g)===CO2(g)+2H2O(g)ΔH=-890.3kJ/mol
D.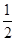 C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l)ΔH=-1400kJ/mol
C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l)ΔH=-1400kJ/mol
湖北省互联网违法和不良信息举报平台 | 网上有害信息举报专区 | 电信诈骗举报专区 | 涉历史虚无主义有害信息举报专区 | 涉企侵权举报专区
违法和不良信息举报电话:027-86699610 举报邮箱:58377363@163.com