
5.3.6g碳在一定量的氧气中燃烧,反应后生成的气体质量为9.2g。则在标准状况下,生成物CO和CO2的体积比为( C )
A.3:2 B.4:1 C.5:1 D.7:4
4.在等体积的NaCl、MgCl2、AlCl3三种溶液中,分别加入等量的AgNO3溶液,恰好都完全反应,则以上三种溶液的物质的量浓度之比为( D )
A.1:2:3 B.3:2:1 C.1:1:1 D.6:3:2
3.将5mol/L的Mg(NO3)2溶液a mL稀释至b mL,稀释后溶液中NO3-的物质的量浓度为( D )
A.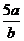 mol/L B.
mol/L B.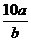 mol/L C.
mol/L C.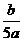 mol/L D.
mol/L D.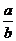 mol/L
mol/L
2.在反应3Cu2S+22HNO3 6Cu(NO3)2+3H2SO4+10NO+8H2O中,1mol还原剂失去原子的物质的量为( C )
6Cu(NO3)2+3H2SO4+10NO+8H2O中,1mol还原剂失去原子的物质的量为( C )
A.6mol B.8mol C.10mol D.12mol
1.与8g SO3中氧原子数相同的是(B )
A.9.6g H2SO4 B.6.6g CO2
C.6.4g SO4 D.6.72L CO(常温、常压)
第二节 书面表达(满分30分)
假设你是李华,你的外国网友Tom最近从网上了解到有关中国的“留守学生(leftbehind students)”的问题,但不是很明白,为此,他通过电子邮件向你询问。请根据下列要点用英语给Tom回一封120词左右的电子邮件:
“留守学生”产生的原因:父母外出打工,无法照顾子女;
“留守学生”遇到的现实问题;
你对解决“留守学生”问题的看法。
Dear Tom,
I am very glad that you asked me the question about the problem with the “leftbehind students” in China.
________________________________________________________________________
________________________________________________________________________
Best regards.
Li Hua
第一节 短文改错(共10小题;每小题1分,满分10分)
(宁波四中2009-2010学年度期中考试卷)
It is five years now since I graduate from No.3 High School.Last Saturday,the class that I was in held a gettogether,which took us a long time prepare.It was indeed not easy to get in the touch with everybody and set a well time for all of us.We all enjoyed this precious day greatly,remember the time we spent together and the people they were familiar with.It was a pity which some of us were not present as they had gone to abroad for further studies,and they called back or sent greeting card from different places.
湖北省互联网违法和不良信息举报平台 | 网上有害信息举报专区 | 电信诈骗举报专区 | 涉历史虚无主义有害信息举报专区 | 涉企侵权举报专区
违法和不良信息举报电话:027-86699610 举报邮箱:58377363@163.com